Abstract
An inspection‑ready QC laboratory relies on structured GMP systems, disciplined documentation, and proactive quality controls to ensure consistent regulatory readiness. Quality Control (QC) laboratories represent the scientific backbone of pharmaceutical manufacturing. Regulatory authorities worldwide—including the U.S. Food and Drug Administration, European Medicines Agency, and World Health Organization—consider laboratory data the ultimate evidence demonstrating that drug products meet predefined specifications for identity, purity, strength, and quality.
During regulatory inspections, QC laboratories receive particular scrutiny because they generate the analytical evidence used to release products to the market. Inspectors evaluate laboratory documentation, analytical methods, equipment calibration, data integrity practices, training systems, and the overall scientific robustness of the laboratory operations.
Inspection readiness is therefore not simply a state achieved days before an audit; it is an ongoing operational philosophy embedded within the laboratory quality system.
This article provides a comprehensive guide for pharmaceutical professionals on preparing QC laboratories for regulatory inspections. It explores regulatory expectations, common inspection findings, data integrity requirements, documentation practices, analytical method controls, and practical strategies to build a laboratory culture capable of sustaining compliance.

The Role of QC Laboratories in Pharmaceutical Compliance
Quality control laboratories perform testing and verification activities that ensure drug products comply with regulatory requirements before release to the market.
Typical responsibilities include:
- Raw material testing
- In-process control testing
- Finished product testing
- Stability studies
- Environmental monitoring
- Method validation
- Investigation of analytical deviations
Regulatory authorities consider QC laboratories critical because the analytical results they produce directly influence product release decisions.
According to regulatory inspection guidance, QC laboratories must demonstrate:
- scientifically sound analytical methods
- documented procedures and specifications
- proper equipment calibration
- reliable data recording practices
- investigation of analytical failures
Inspectors typically assess laboratory operations through direct observation of testing procedures and review of raw analytical data to verify compliance with Good Manufacturing Practices (GMP).
Structure of a Modern Pharmaceutical QC Laboratory
A typical pharmaceutical QC laboratory is organized into specialized analytical units.
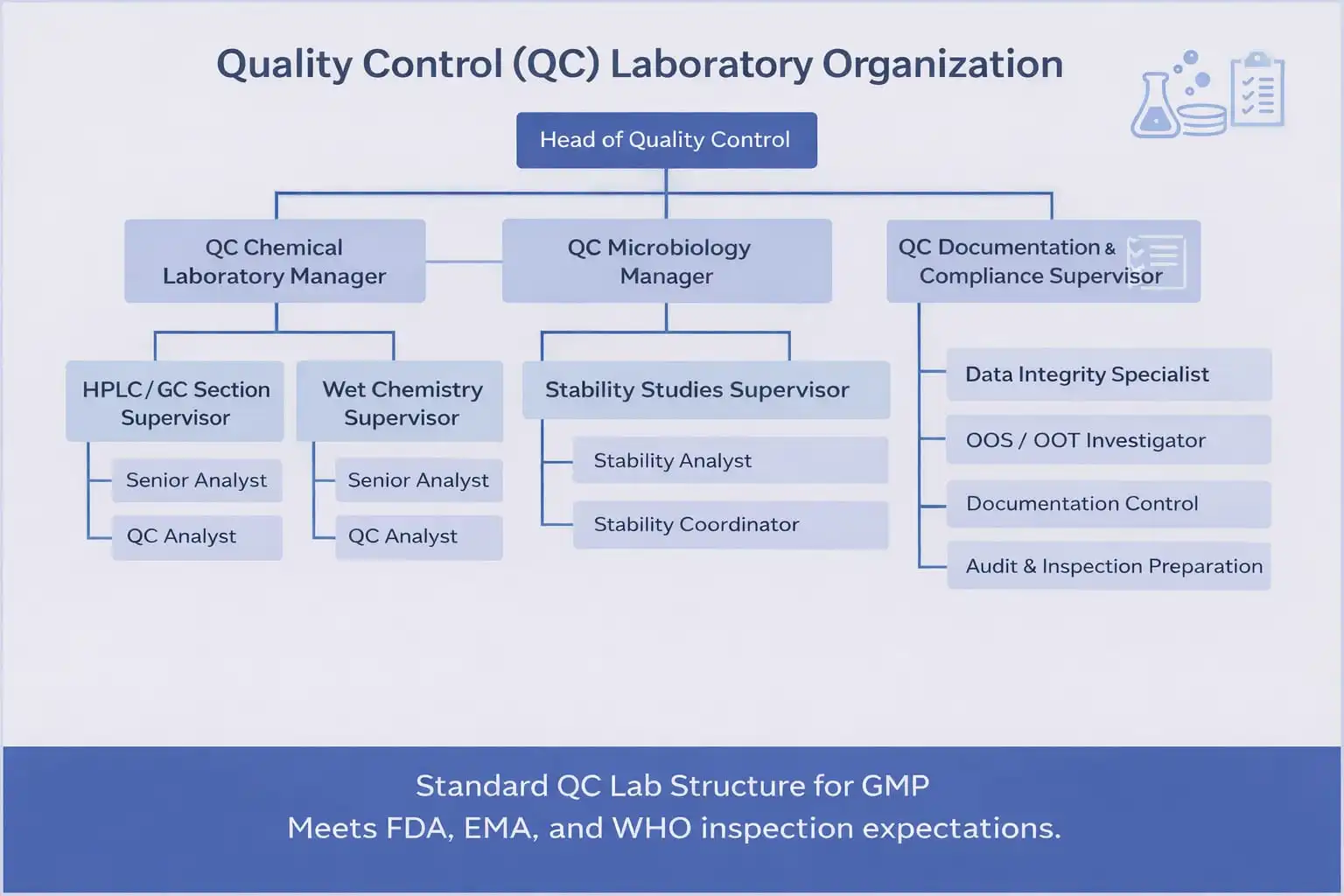
Common laboratory sections include:
1-Physicochemical Analysis Unit
Responsible for techniques such as:
- HPLC
- GC
- UV spectroscopy
- Titration
- Dissolution testing
2-Microbiology Laboratory
Responsible for:
- sterility testing
- endotoxin testing
- microbial limits
- environmental monitoring
3-Stability Laboratory
Conducts long-term and accelerated stability studies according to ICH guidelines.
4-Raw Material Testing Unit
Ensures incoming materials meet pharmacopoeia or internal specifications.
QC Workflow and Sample Lifecycle
Understanding the sample lifecycle is critical for inspection readiness.
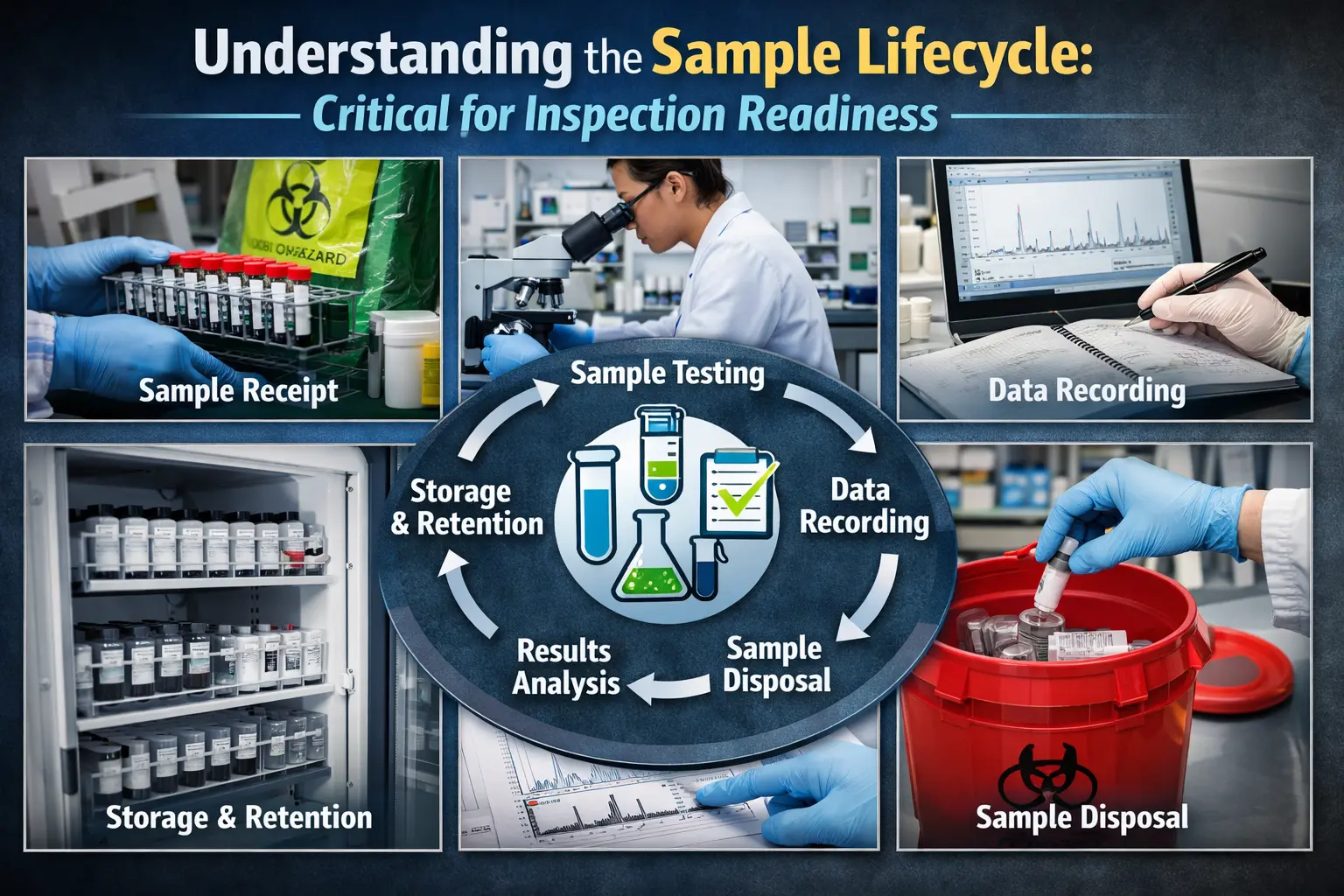
Typical QC sample lifecycle:
- Material receipt
- Sampling according to SOP
- Sample registration
- Laboratory testing
- Data review
- Certificate of analysis generation
- Batch disposition
Each stage must be fully documented.
Inspectors often review sample traceability to confirm:
- samples were properly labeled
- tests were performed according to procedures
- analytical results correspond to batch history
Regulatory Expectations During QC Inspections
Inspectors focus on verifying that laboratory activities comply with Good Manufacturing Practices (GMP).
Authorities such as the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use have established frameworks defining laboratory lifecycle and analytical quality.
Typical inspection focus areas include:
1-Analytical Methods
Inspectors verify that methods are:
- validated
- appropriate for intended use
- documented
2-Equipment Qualification
Laboratory equipment must undergo:
- Installation Qualification (IQ)
- Operational Qualification (OQ)
- Performance Qualification (PQ)
3-Calibration and Maintenance
Instruments must have documented calibration records.
4- Method Validation
Validation parameters include:
- accuracy
- precision
- specificity
- linearity
- robustness
5-Data Integrity
Inspectors review chromatograms, integration parameters, and raw data.
Data Integrity: A Critical Inspection Topic
Data integrity has become the most common reason for regulatory warning letters.
The industry follows the ALOCA+ principles for maintaining trustworthy laboratory data.
ALOCA+ stands for:
- Attributable – who performed the analysis
- Legible – readable records
- Original – original data preserved
- Contemporaneous – recorded at the time of activity
- Accurate – correct results
Additional attributes include:
- Complete
- Consistent
- Enduring
- Available
Data integrity failures often occur due to:
- deleted electronic data
- improper chromatographic integration
- unofficial worksheets
- undocumented retesting
These issues frequently trigger regulatory actions.
Common Inspection Findings in QC Laboratories
Based on regulatory observations worldwide, common inspection findings include:
1-Inadequate OOS Investigations
Out-of-specification results must be investigated scientifically rather than automatically retested.
2-Incomplete Documentation
Examples include:
- missing chromatograms
- undocumented calculations
- incomplete test records
3-Poor Instrument Maintenance
Inspectors often discover instruments used beyond calibration dates.
4-Improper Analytical Practices
Examples:
- undocumented sample dilutions
- incorrect standard preparation
5-Weak Training Systems
Laboratory analysts must be qualified before performing analytical methods.
Documentation: The Backbone of Inspection Readiness
Laboratory documentation provides evidence of compliance.
Key documentation systems include:
- Standard Operating Procedures (SOPs)
- Analytical worksheets
- Equipment logbooks
- Training records
- Investigation reports
Inspectors often compare:
- raw data
- laboratory notebooks
- final reports
Analytical Techniques and Instrument Control
Analytical instruments must operate under controlled conditions.
Common QC instruments include:
- HPLC
- GC
- UV-Vis spectrophotometers
- Dissolution testers
- Karl Fischer titrators
For example, chromatographic methods must demonstrate adequate system suitability before sample analysis.
System suitability parameters often include:
- theoretical plates
- tailing factor
- resolution
- repeatability
Laboratory Investigations and Root Cause Analysis
Analytical failures require systematic investigations.
Root cause analysis tools include:
- Ishikawa diagrams
- Five Whys
- FMEA
The Ishikawa (fishbone) diagram is widely used in pharmaceutical quality systems to identify factors influencing product quality and analytical results.
Investigations should document:
- the event
- potential causes
- corrective actions
- preventive actions
Analyst Qualification and Training
A laboratory is only as reliable as the analysts operating it.
Training programs must include:
- GMP training
- analytical technique training
- instrument operation
- data integrity awareness
Analysts should be qualified through:
- method demonstration runs
- supervised testing
- competency evaluation
Training records are commonly reviewed during inspections.
Building an Inspection-Ready QC Laboratory
Inspection readiness requires proactive quality culture.
Key elements include:
1-Internal Audits
Routine laboratory self-inspections help identify compliance gaps.
2-Mock Inspections
Simulated inspections prepare analysts for regulatory interviews.
3-Risk-Based Quality Systems
Risk assessments help prioritize critical processes.
4-Continuous Training
Analysts should regularly update their technical knowledge.
5-Strong Quality Oversight
Quality Assurance should review laboratory operations periodically.
Future Trends in QC Inspection Readiness
Modern pharmaceutical laboratories are moving toward digital transformation.
Emerging technologies include:
- electronic laboratory notebooks (ELN)
- laboratory information management systems (LIMS)
- automated chromatography systems
- AI-assisted data analysis
These technologies improve:
- traceability
- data integrity
- efficiency
However, digital systems must still comply with regulatory requirements such as electronic record controls.
LABORATORY MANAGEMENT
Overall management of the laboratory work, its staff, and the evaluation of the results of analysis are important elements in the evaluation of a control laboratory. Span of supervisory control, personnel qualifications, turnover of analysts, and scope of the laboratory’s responsibility are important issues to examine when determining the quality of overall management and supervision of work. Individually or collectively, these factors are the basis for an objection only when they are shown to result in inadequate performance of responsibilities required by the CGMPs.
Review laboratory logs for the sequence of analysis and the sequence of manufacturing dates. Examine laboratory records and logs for vital information about the technical competence of the staff and the quality control procedures used in the laboratory.
Observe analysts performing the operations described in the application. There is no substitute for actually seeing the work performed and noting whether good technique is used. You should not stand over the analysts, but watch from a distance and evaluate their actions.
Sometimes the company’s employees have insufficient training or time to recognize situations that require further investigation and explanation. Instead they accept unexplained peaks in chromatograms with no effort to identify them. They may accept stability test results showing an apparent increase in the assay of the drug with the passage of time with no apparent question about the result. Also, diminishing reproducibility in HPLC chromatograms appearing several hours after system suitability is established is accepted without question.
Good manufacturing practice regulations require an active training program and the documented evaluation of the training of analysts.
The authority to delete files and override computer systems should be thoroughly examined. Evaluate the history of changes to programs used for calculations. Certain changes may require management to re-examine the data for products already released.
Conclusion
QC laboratories represent one of the most critical elements of pharmaceutical manufacturing compliance. Regulatory authorities rely heavily on laboratory data when evaluating product quality and safety.
Inspection readiness requires more than compliance with written procedures—it requires a laboratory culture that prioritizes scientific integrity, accurate documentation, and continuous improvement.
Organizations that invest in strong laboratory quality systems, robust data integrity practices, and well-trained analysts significantly reduce the risk of regulatory findings.
Ultimately, the goal of inspection readiness is not merely to satisfy regulators but to ensure that every batch of medicine released to patients meets the highest standards of quality and safety.
References
U.S. Food and Drug Administration – Guide to Inspections of Pharmaceutical Quality Control Laboratories
World Health Organization – good practices for pharmaceutical quality control laboratories
International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use – Quality Guidelines
European Medicines Agency – GMP Compliance Guidance
U.S. Food and Drug Administration – Guide to Inspections of Pharmaceutical Quality Control Laboratories
EDA accreditation of quality control laboratories of pharmaceutical companies program


