1.Introduction
1.1 What are Nitrosamines
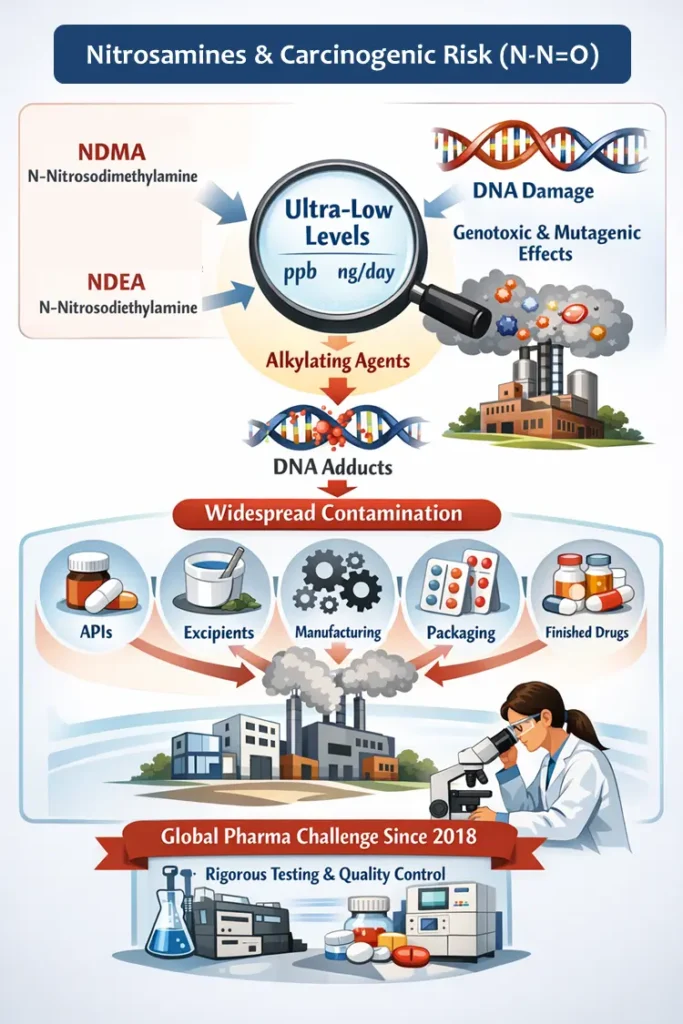
Nitrosamines are a class of organic compounds containing a nitroso functional group (–N–N=O). Many nitrosamines, including N-nitrosodimethylamine (NDMA) and N-nitrosodiethylamine (NDEA), are classified as probable human carcinogens because of their genotoxic and mutagenic properties. Mechanistically, nitrosamines exert carcinogenicity through metabolic activation, producing highly reactive alkylating species that form DNA adducts. The affecting concentration is usually at ultra-low levels, often in parts-per-billion (ppb) or nanogram-per-day, which underscores the need to control and monitor these impurities through highly sensitive analytical systems, process controls, and quality systems. Since 2018, nitrosamine impurities have emerged as one of the most consequential quality and patient-safety challenges facing the global pharmaceutical industry. What initially appeared to be an isolated active pharmaceutical ingredient (API)–related issue rapidly evolved into a systemic risk spanning APIs, excipients, manufacturing processes, packaging, and finished drug products.
1.2 Why nitrosamines matter?
The nitrosamines issue was first brought into global attention in 2018, although the reaction is not new, rather pretty old, i.e., since 1970. The unprecedented triggering of nitrosamines called for regulatory actions, risk assessment, and profound knowledge of material versus process. Since its inception, there have been large-scale recalls across major pharmaceutical markets. These events revealed critical realities that continue to shape regulatory thinking. For instance, nitrosamine risk is not confined to a single product, manufacturer, or process step; therefore, patient safety implications are significant, particularly given the chronic exposure potential of many affected medicines. Currently, traditional impurity control paradigms are insufficient for ultra-potent genotoxic impurities. As a result, nitrosamines have transitioned from a niche quality topic to a board-level strategic priority, which influences product design, supplier qualification, lifecycle management, and regulatory engagement across the industry.

1.3 Formation mechanism
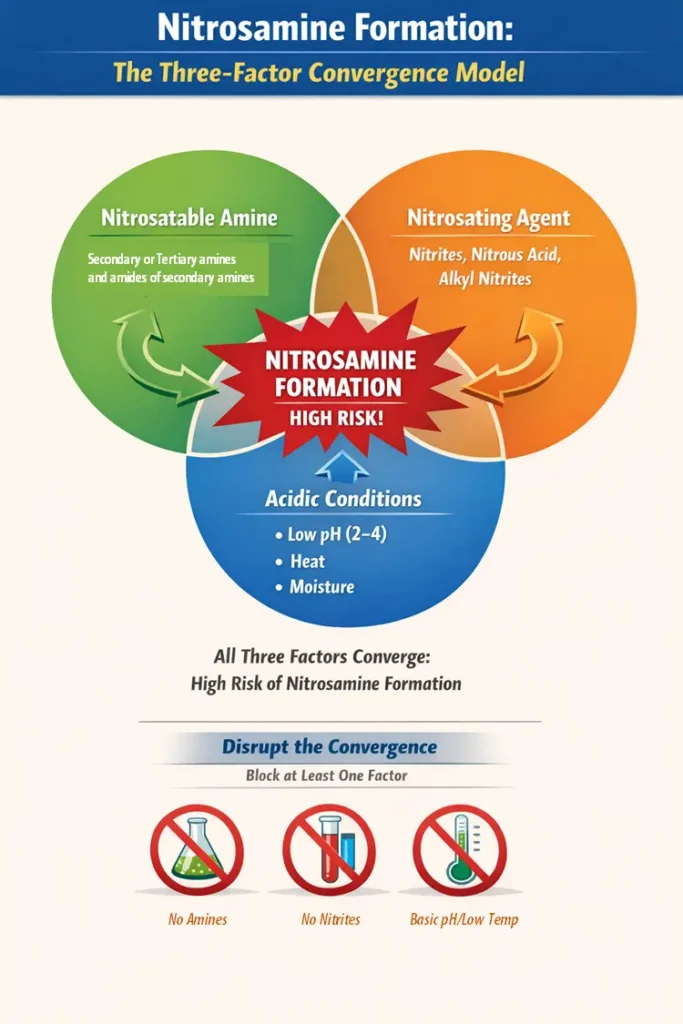
The formation mechanism of nitrosamines consists of a chemical reaction called nitrosation. It is a reaction in which an amine, which may be nitrosatable, undergoes nitrosation in the presence of a nitrosating agent, such as nitrites, nitrous acid, alkyl nitrites, under conducive acidic conditions. Usually, secondary amines, tertiary amines (via rearrangement), and amides of secondary amines are particularly susceptible to this reaction. Basically, this reaction occurs when three key elements converge, such as a nitrosatable amine, a nitrosating agent (e.g., nitrites, nitrous acid, alkyl nitrites), and favorable conditions, typically acidic pH (2–4), elevated temperature, and the presence of water or moisture. The formation mechanism can be viewed as a three-factor convergence model. When all three elements coexist, the probability of formation increases exponentially; therefore, deliberately disrupting at least one element of this convergence is imperative to effectively control nitrosamine formation.
1.4 Predictive assessment
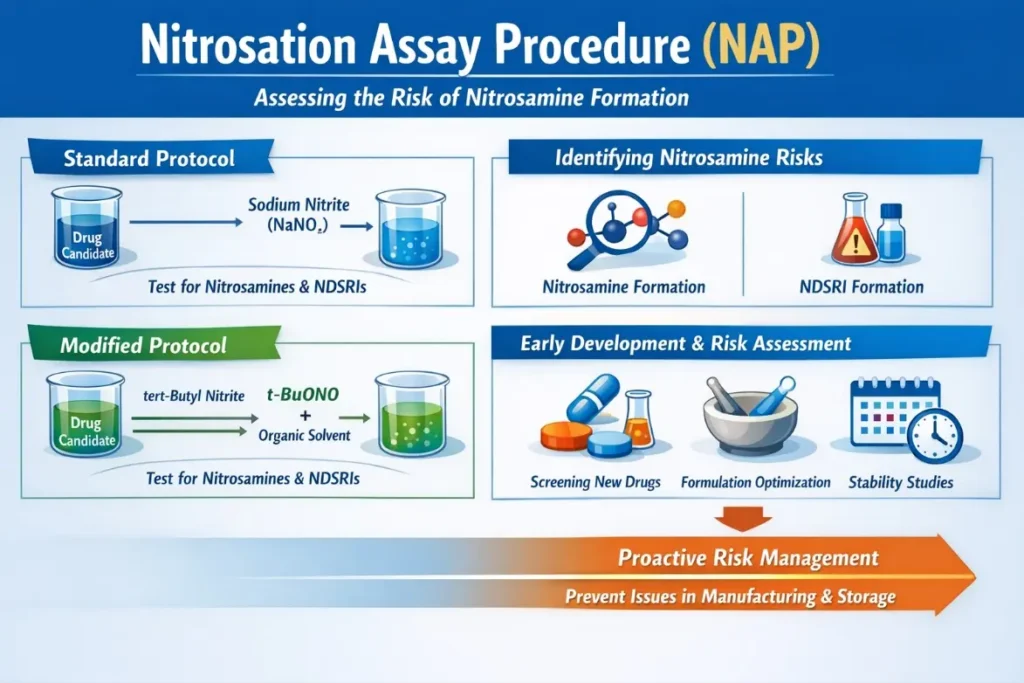
The Nitrosation Assay Procedure (NAP) is an in-vitro screening tool designed to assess the propensity of drug substances or excipients to form nitrosamines or nitrosamine drug-substance-related impurities (NDSRIs). Standard protocols employ sodium nitrite in aqueous systems, whereas modified protocols utilize organic solvents and tert-butyl nitrite to improve solubility and reactivity. These assays are increasingly embedded into early development programs and lifecycle risk assessments, supporting proactive identification of nitrosamine risks before they manifest during commercial manufacture or stability storage.
2.Common risk sources in pharmaceutical manufacturing
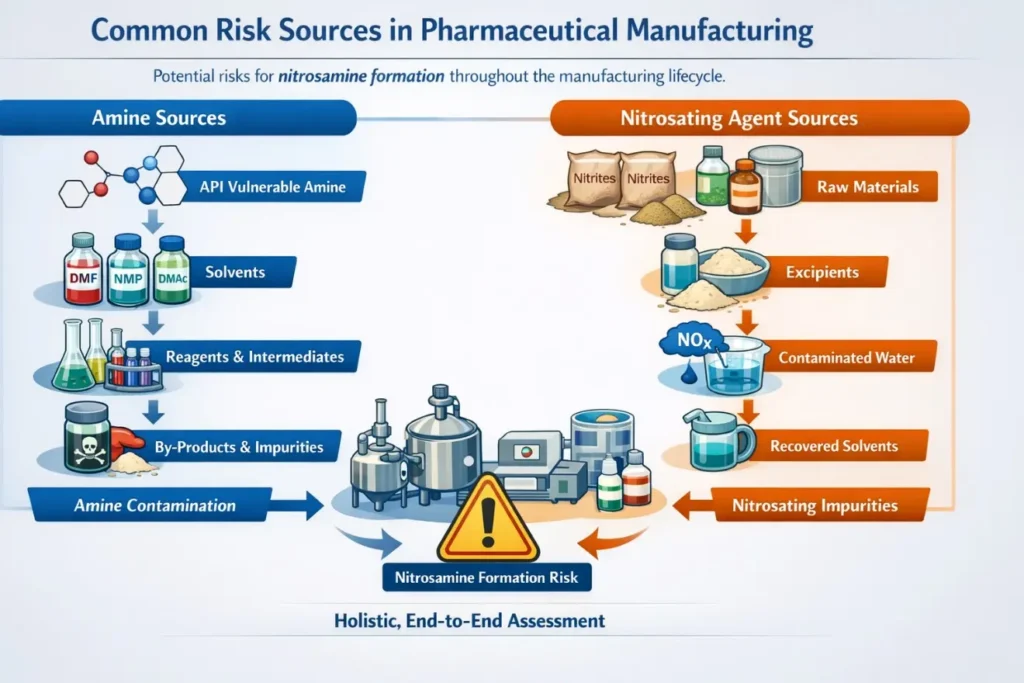
Common risk sources in pharmaceutical manufacturing for nitrosamine formation are numerous, spanning the manufacturing lifecycle, and require holistic, end-to-end assessment strategies. Generally, amines and nitrosating agents are the main sources.
2.1 Amine sources
There can be numerous amine sources, for instance, the actual API molecule, which may contain a vulnerable amine within its structure and which may be prone to nitrosation. Presence of solvents such as dimethylformamide (DMF), N-methylpyrrolidone (NMP), and dimethylacetamide (DMAc), which may contain or generate amines, could be one of the sources of amines. In addition, reagents, intermediates, and by-products during manufacturing steps can introduce amine impurities.
2.2 Nitrosating agent sources
The major risk factor, given the sources of nitrosating agents, is impurities in the raw materials, especially nitrites in the starting materials. Also, excipients may contain trace amounts of nitrites or nitrates, which could lead to impurity formation downstream. There is also a risk of contamination of water systems and recovered solvents with nitrogen oxides, nitrites, or nitrates, which add to the risk profile. The risk vectors often overlap across various levels of the manufacturing process, which is why a holistic, end-to-end risk assessment and control approach is crucial.
3.Famous case studies (Chosen ones)
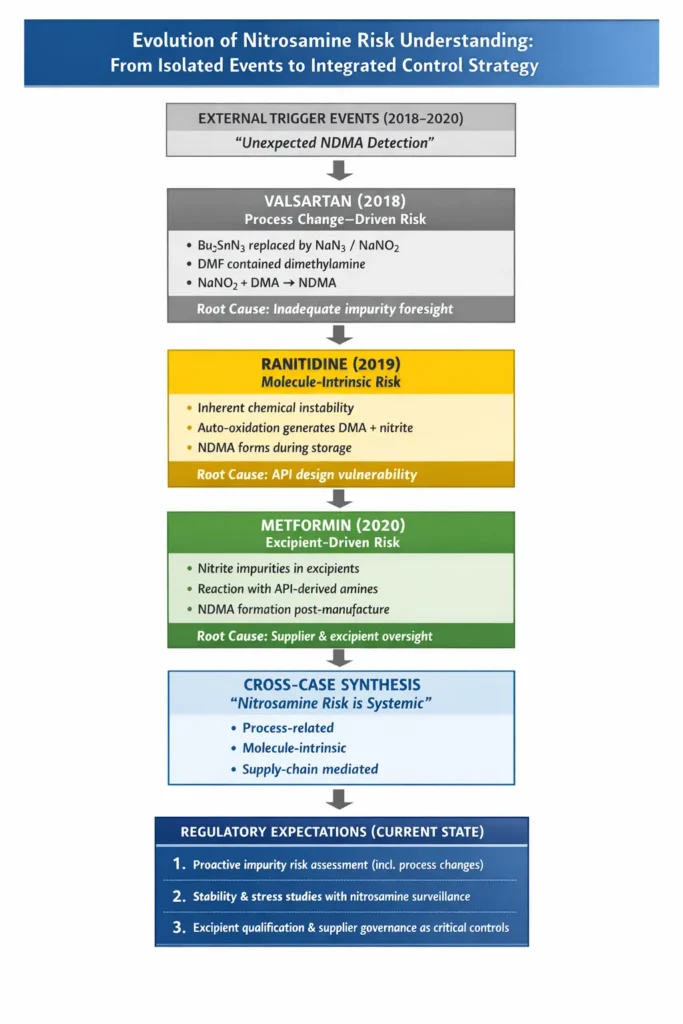
3.1 Valsartan case – The first reported case in 2018
In the valsartan case, a manufacturing process modification replaced tributyltin azide (Bu3SnN3) with sodium azide and sodium nitrite (NaN3). While operationally efficient, this change introduced a critical interaction between sodium nitrite and dimethylamine, which was present in DMF, and resulted in NDMA formation. The lesson was unequivocal: even minor process changes demand comprehensive impurity risk assessments with a forward-looking lens.
3.2 Ranitidine (2019): Intrinsic Molecular Instability
The Ranitidine case represented a paradigm shift. NDMA formation was linked to inherent molecular instability, with the drug substance degrading via auto-oxidation to generate both dimethylamine and nitrite under normal and accelerated conditions. This demonstrated that nitrosamine risk can originate from the drug substance itself, necessitating enhanced stability studies and stress-condition monitoring.
3.3 Metformin (2020): Excipients as Active Contributors
In the metformin case, NDMA formation was attributed to nitrite impurities in excipients reacting with the API. This case dismantled the long-held assumption that excipients are passive ingredients, but can be critical contributors of nitrite. This underscored excipient qualification, nitrite specification, and supplier governance at critical control points.
3.4 Key lessons learnt
Across these cases, three high-value lessons have firmly been embedded into regulatory expectations:
1. Process changes must be supported by robust impurity risk assessments.
2. Stability testing must include stress conditions and nitrosamine monitoring.
3. Excipient quality, solvent selection, and supplier controls are non-negotiable.
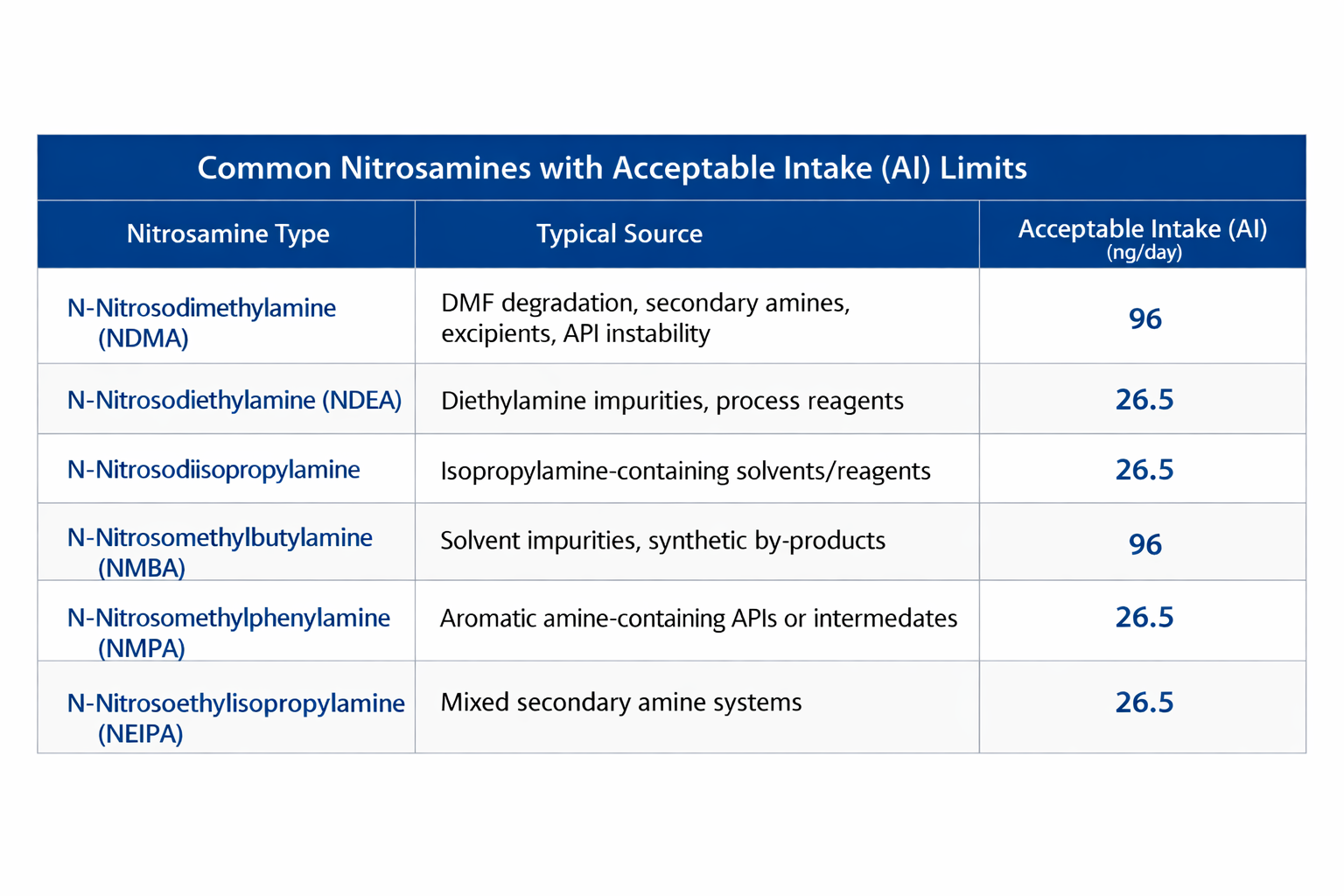
4.Common nitrosamines with acceptable intake (AI) limits
Several nitrosamines have been most frequently observed across pharmaceutical products. Current FDA acceptable intake (AI) limits are summarized in Table. Importantly, these limits apply on a per-day exposure basis, not per unit dose. Consequently, high-dose drug products face disproportionately stringent ppb-level specifications, an operational reality that directly drives analytical strategy, formulation design, and risk mitigation approaches.
5.Conclusion
Overall, the quality risk caused by nitrosamines is a systemic and multifactorial phenomenon that stretches much further than single manufacturing processes. The historical lessons taught on the need to take proactive, science-based risk management that is integrated in the design of the product, supply chains, and lifecycle governance. In the future, sustainable compliance will require anticipatory assessment, cross-functional responsibility, and changing reactive controls to predictive strategies of quality control.
Note: This article represents the first in a structured series on nitrosamines. The intent of this opening part is to establish a foundational understanding of nitrosamines. For instance, what nitrosamines are, how they form, where risks originate, and why several historical cases fundamentally reshaped global regulatory expectations. Subsequent articles will build on this foundation, addressing analytical strategies, mitigation approaches, regulatory frameworks, and future risk models.
References
- Control of nitrosamine impurities in human drugs – FDA
- CDER Nitrosamine Impurity Acceptable Intake Limits
- Good practice considerations for preventing and controlling nitrosamines – WHO
- Nitrosamine impurities in medicines – TGA
- Guidance on nitrosamine impurities in medications – Health Canada
- Recommended Acceptable Intake Limits for Nitrosamine Drug Substance-Related Impurities (NDSRIs) – FDA
- Assessment and Control of DNA Reactive (Mutagenic) Impurities in Pharmaceuticals to Limit Potential Carcinogenic Risk M7(R2) – ICH