Finale Introduction: When Medicine Stops Guessing
In this concluding section, we delve into the transformative journey of electrogenomics, tracing its evolution from a mere concept to a tangible reality that intertwines polymer chemistry with the intricacies of intracellular decision-making. As we stand on the brink of a new era in medicine, we are compelled to confront critical questions that will shape the future of healthcare: Can we build this technology responsibly?
What frameworks can ensure its regulation? Is it scalable to meet the demands of diverse populations? Most importantly, what implications arise when medicine shifts from a reactive approach, waiting for symptoms, to a proactive one that responds directly to gene expression? This pivotal moment presents a crossroads where the future of healthcare can either solidify into a robust infrastructure or remain an elusive fiction.

In this article, we explore the remarkable evolution of electrogenomics, a field that merges polymer chemistry with the complexities of intracellular decision-making. As we stand on the cusp of a new era in medicine, we are faced with critical questions that will shape the future of healthcare. This exploration not only highlights the potential of this technology but also emphasizes the ethical, regulatory, and scalability challenges that accompany its integration into medical practice. By examining these pivotal issues, we aim to illuminate the path forward, ensuring that the transition from reactive to proactive healthcare is both responsible and effective.
Electrogenomics represents a groundbreaking intersection of biology and technology, where the principles of polymer chemistry are harnessed to influence cellular behavior at the genetic level. This innovative approach has evolved from a theoretical concept into a practical reality, promising to revolutionize how we understand and treat diseases. As we delve deeper into this transformative journey, it becomes evident that we are not merely observing a scientific advancement; we are witnessing a paradigm shift in healthcare.
The potential of electrogenomics to proactively address health issues by directly interacting with gene expression raises significant questions. Can we develop this technology in a manner that prioritizes safety and ethical considerations? What regulatory frameworks are necessary to ensure its responsible use? Furthermore, as we consider the scalability of this technology, we must ask whether it can be adapted to meet the diverse needs of various populations.
At the heart of this discussion lies a fundamental shift in the approach to medicine. Traditionally, healthcare has been reactive, focusing on treating symptoms after they arise. Electrogenomics, however, offers the possibility of a proactive model, where interventions can be tailored to individual genetic profiles before symptoms manifest. This shift not only has the potential to enhance patient outcomes but also challenges us to rethink our current healthcare infrastructure.
As we navigate this pivotal moment, we stand at a crossroads. The decisions we make today will determine whether the future of healthcare solidifies into a robust and equitable system or remains an elusive vision. By addressing the ethical, regulatory, and practical implications of electrogenomics, we can pave the way for a new era of medicine that is both innovative and responsible.
Regulatory Reality: Combination Products Are the Blueprint
In the evolving landscape of medical technology, electrogenomic systems represent a significant advancement that challenges traditional regulatory frameworks. This document explores how these systems, which integrate drugs, devices, and biologics, are not merely standalone entities but rather fall under the category of FDA combination products. By examining existing regulatory precedents and the nuances of hybrid frameworks, we can better understand how electrogenomics fits into the current regulatory paradigm.
Understanding Combination Products
Combination products are defined by the FDA as therapeutic and diagnostic products that combine drugs, devices, and/or biological products. This regulatory classification is crucial for electrogenomic systems, which inherently blend these categories. The FDA’s approach to combination products allows for a more streamlined evaluation process, ensuring that innovative technologies can reach the market while maintaining safety and efficacy standards.
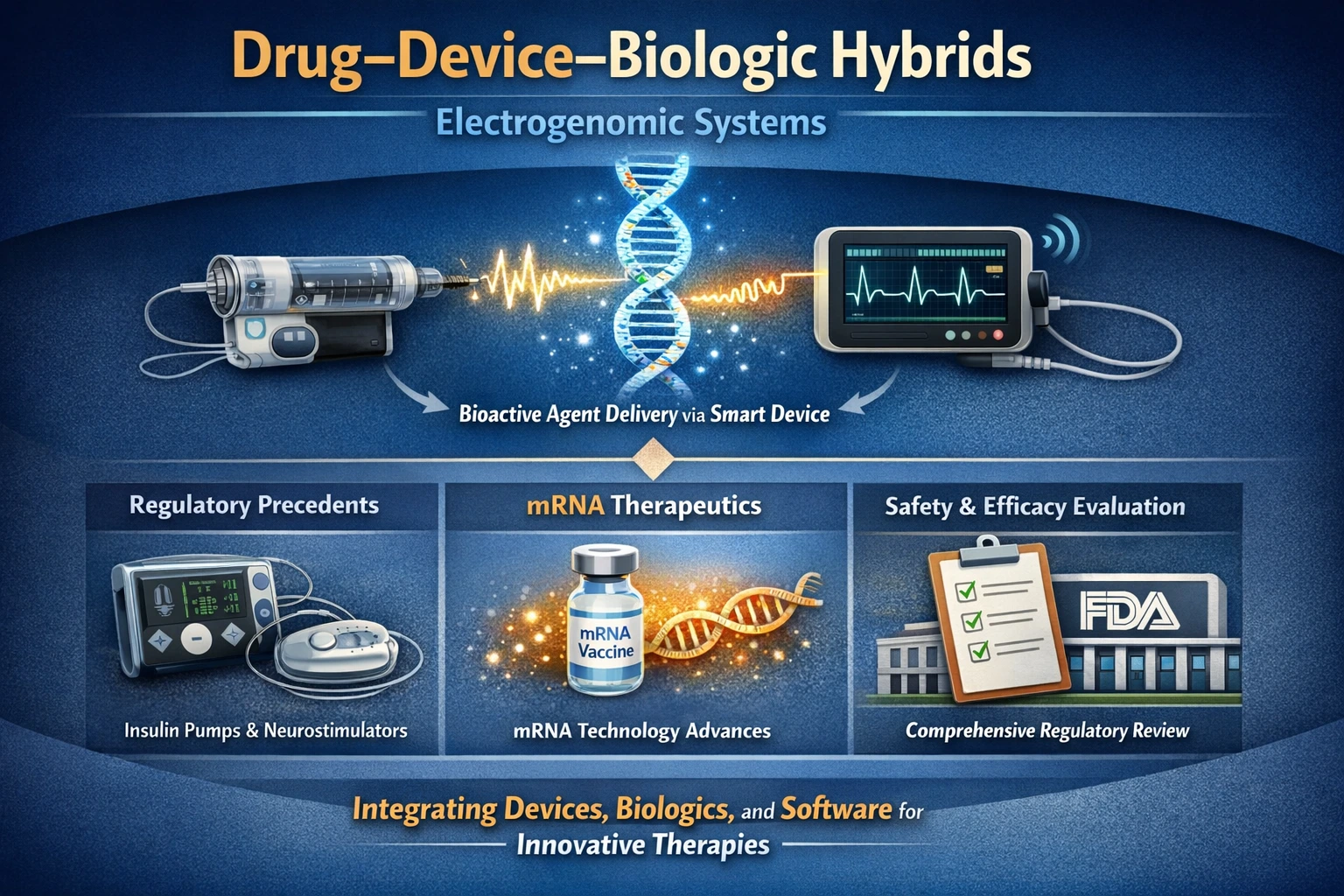
Key Components of Combination Products
- Drug–Device–Biologic Hybrids: Electrogenomic systems often incorporate elements from all three categories. For instance, a system may deliver a biologic agent through a device that is governed by software algorithms, thus necessitating a comprehensive regulatory review.
- Existing Precedents: The FDA has already established frameworks for products like insulin pumps and neurostimulators, which serve as valuable reference points. These products have successfully navigated the regulatory landscape, demonstrating that hybrid systems can be effectively evaluated for safety and efficacy.
- mRNA Therapeutics: The recent surge in mRNA-based therapies has further illustrated the FDA’s adaptability in regulating innovative products. These therapies often involve complex interactions between biological components and delivery mechanisms, similar to electrogenomic systems.
Regulatory Evaluation of Electrogenomic Systems
The regulatory evaluation of electrogenomic systems involves several critical aspects that align with existing frameworks for combination products.
Signal-Triggered Actuation
Electrogenomic systems often utilize signal-triggered actuation, where external stimuli activate the delivery of therapeutic agents. This mechanism requires careful evaluation to ensure that the system responds accurately and safely to the intended signals. Regulatory bodies will assess the reliability of these triggers and their potential impact on patient outcomes.
Implantable Polymers
Many electrogenomic systems incorporate implantable polymers that serve as drug delivery vehicles. The biocompatibility and degradation profiles of these materials are essential considerations in the regulatory process. The FDA will evaluate how these polymers interact with biological systems and their long-term effects on patients.
Software-Governed Thresholds
Software plays a pivotal role in the operation of electrogenomic systems, particularly in determining thresholds for therapeutic intervention. Regulatory agencies will scrutinize the algorithms and software used to ensure they meet safety standards and function as intended. This includes assessing the potential for software updates and the implications for ongoing patient care.
Extending Regulatory Physics
Electrogenomics does not disrupt the existing regulatory framework; rather, it extends the principles already in place. By leveraging the lessons learned from previous combination products, regulators can apply established methodologies to evaluate these innovative systems effectively.
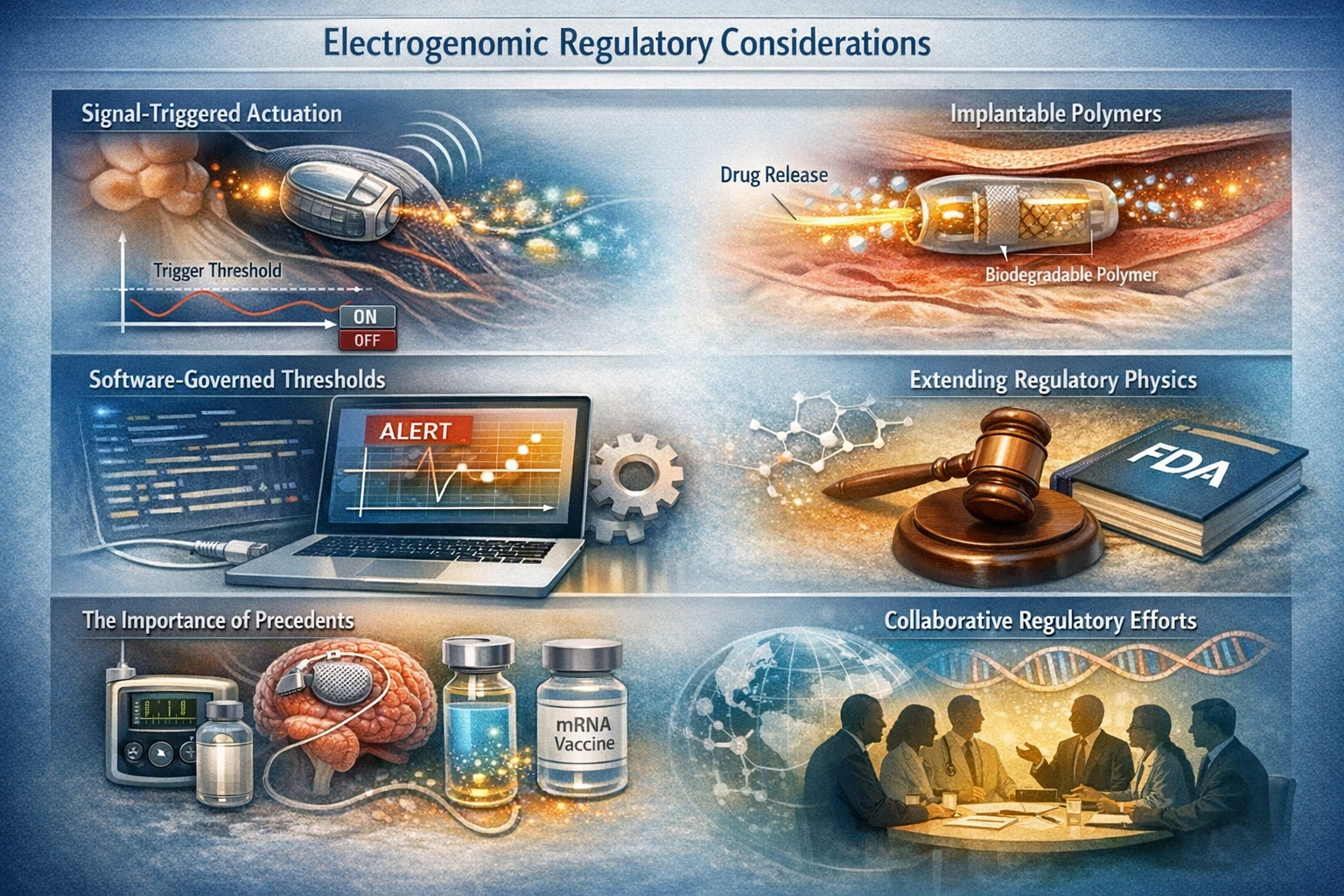
The Importance of Precedents
The success of insulin pumps, neurostimulators, and mRNA therapeutics provides a roadmap for the regulatory evaluation of electrogenomic systems. These precedents highlight the importance of a flexible regulatory approach that accommodates the complexities of hybrid technologies.
Collaborative Regulatory Efforts
As the field of electrogenomics continues to evolve, collaboration between regulatory agencies, industry stakeholders, and researchers will be essential. Engaging in dialogue and sharing insights can help shape a regulatory environment that fosters innovation while ensuring patient safety.
Electrogenomic systems represent a frontier in medical technology that necessitates a nuanced understanding of regulatory frameworks. By recognizing these systems as combination products, regulators can apply existing precedents and methodologies to ensure their safe and effective integration into clinical practice. As we move forward, it is crucial to embrace the complexities of these hybrid technologies while maintaining a commitment to patient safety and efficacy. The regulatory reality is clear: combination products are not just a category; they are the blueprint for the future of medical innovation.
Manufacturing Landscape: Difficult, But Not Impossible
The manufacturing landscape for pharmaceuticals is evolving, presenting both challenges and opportunities. As the industry shifts towards more complex systems, the integration of advanced technologies and methodologies becomes essential. This document explores the realities of modern manufacturing in the pharmaceutical sector, emphasizing the alignment with semiconductor biology, the importance of microfabrication, and the need for innovative approaches to ensure consistency and compliance.
Manufacturing Realities
Microfabrication and Polymer Batch Consistency
Microfabrication techniques are increasingly being adopted in pharmaceutical manufacturing. These methods, traditionally associated with semiconductor production, allow for the precise control of material properties and dimensions at the microscale. This precision is crucial for the development of advanced drug delivery systems and personalized medicine.
However, achieving batch consistency in polymer manufacturing remains a significant challenge. Variability in polymer properties can lead to inconsistent drug release profiles and affect the overall efficacy of the product. To address this, manufacturers must invest in robust quality control measures and advanced characterization techniques to ensure that each batch meets stringent specifications.
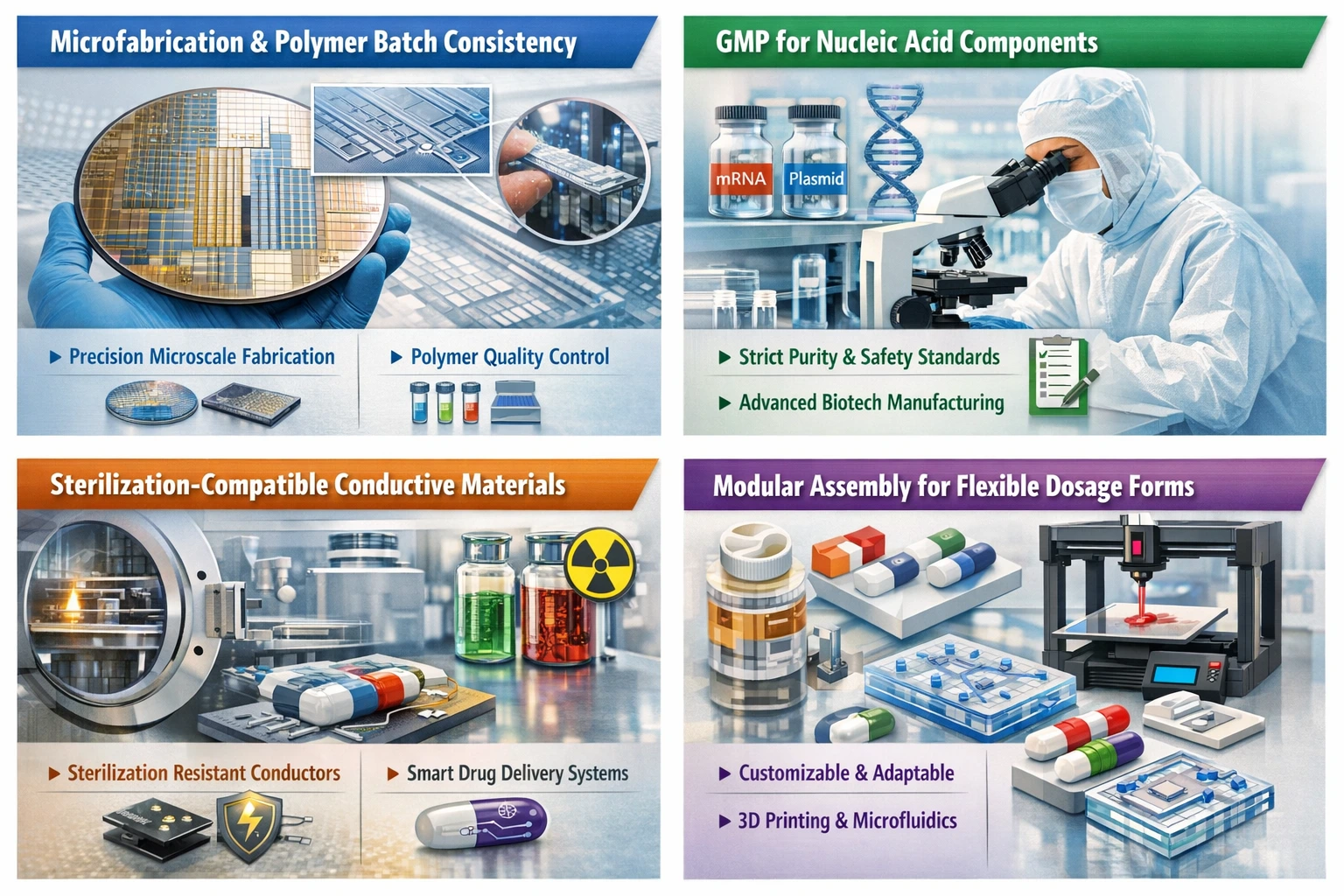
Good Manufacturing Practices (GMP) for Nucleic Acid Components
As the pharmaceutical industry embraces biologics and gene therapies, the need for stringent Good Manufacturing Practices (GMP) for nucleic acid components becomes paramount. The production of plasmids, mRNA, and other nucleic acid-based therapies requires specialized facilities and processes to ensure purity, potency, and safety.
Implementing GMP in this context involves meticulous attention to detail, from raw material sourcing to final product testing. Manufacturers must navigate regulatory requirements while also adopting innovative technologies that enhance efficiency and reduce the risk of contamination.
Sterilization-Compatible Conductive Materials
The integration of conductive materials in pharmaceutical manufacturing processes introduces additional complexities, particularly concerning sterilization. Many conductive materials are not inherently compatible with standard sterilization methods, which can compromise product integrity.
To overcome this challenge, researchers and manufacturers are exploring new sterilization-compatible conductive materials. These materials must maintain their electrical properties while also withstanding the rigors of sterilization processes such as autoclaving or gamma irradiation. The development of such materials is crucial for the advancement of smart drug delivery systems and other innovative therapeutic modalities.
Modular Assembly Rather Than Monolithic Dosage Forms
The traditional approach to pharmaceutical manufacturing often involves the creation of monolithic dosage forms, which can be inflexible and difficult to adapt to changing market demands. In contrast, modular assembly offers a more agile manufacturing paradigm, allowing for the rapid assembly of diverse dosage forms tailored to specific patient needs.
This shift towards modularity not only enhances flexibility but also facilitates the integration of advanced technologies such as 3D printing and microfluidics. By adopting a modular approach, manufacturers can streamline production processes, reduce lead times, and respond more effectively to emerging trends in personalized medicine.
Use Cases That Feel Like Science Fiction (But Are Already Real)
In the realm of modern medicine, several innovative technologies and approaches are emerging that seem to belong in the pages of science fiction. However, these advancements are not merely speculative; they are already being implemented in clinical settings. This document explores five groundbreaking use cases that leverage advanced biological systems to address complex medical challenges, including cancer treatment, autoimmunity management, neuroinflammation, transplant rejection monitoring, and metabolic disease management. Each case illustrates how integration of existing technologies can lead to significant improvements in patient outcomes.
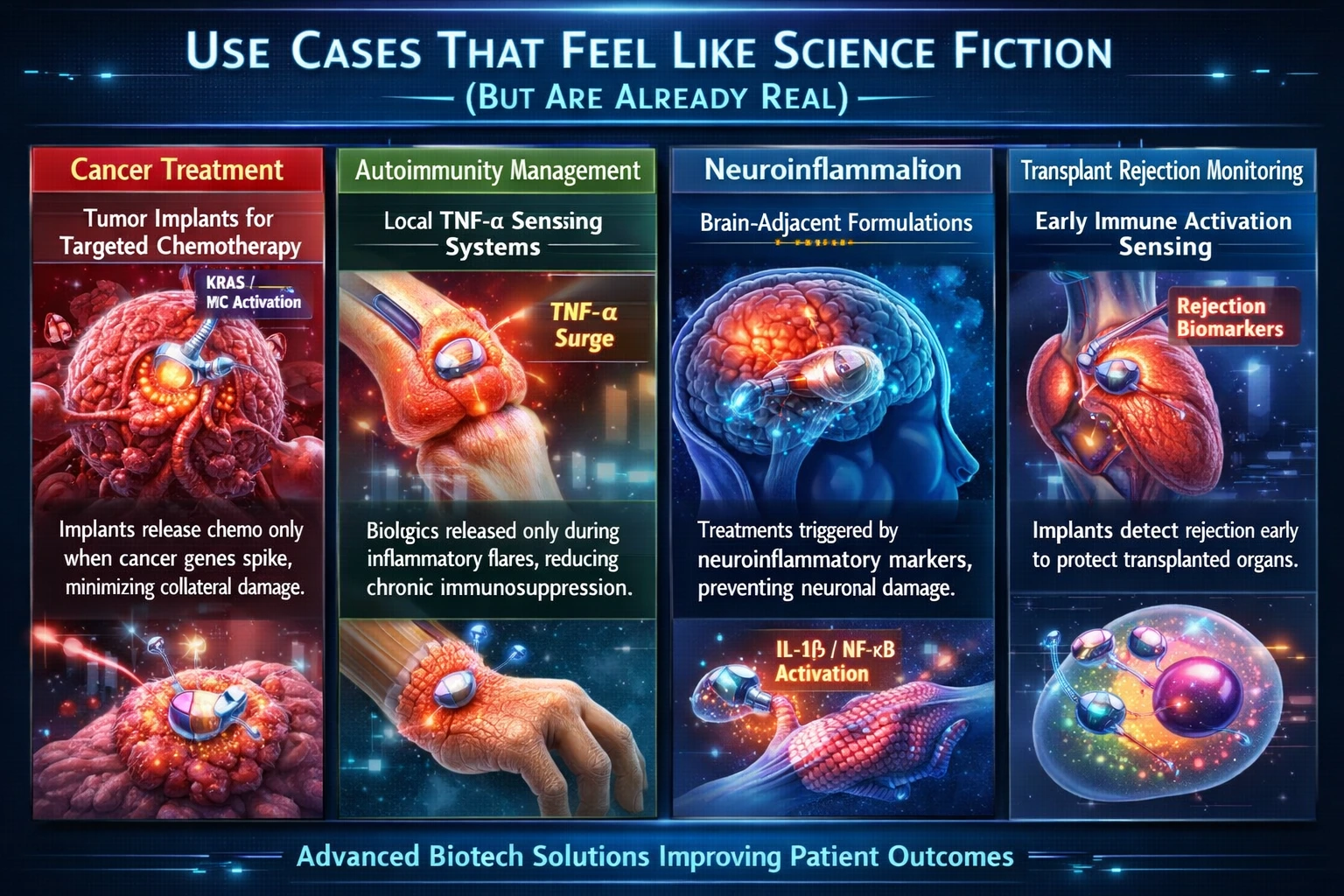
Cancer Treatment
Tumor Implants for Targeted Chemotherapy
One of the most promising advancements in cancer treatment involves the development of tumor implants that release chemotherapy agents only when specific genetic markers, such as KRAS or MYC transcripts, spike. This targeted approach aims to prevent the cycles of resistance that often occur with traditional chemotherapy, as well as minimize collateral toxicity to healthy tissues. By monitoring the molecular signals associated with tumor activity, these implants can deliver treatment precisely when it is needed, enhancing efficacy while reducing side effects. This integration of real-time monitoring and localized drug delivery represents a significant leap forward in personalized cancer therapy.
Autoimmunity Management
Local TNF-α Sensing Systems
In the field of autoimmunity, local TNF-α-sensing systems are being developed to release biologics only during inflammatory flares. This approach eliminates the need for chronic immunosuppression, which can lead to increased vulnerability to infections and other complications. By utilizing sensors that detect elevated levels of TNF-α, these systems can activate treatment precisely when the immune system is overreacting, allowing for a more balanced immune response. This targeted delivery not only improves patient safety but also enhances the quality of life for individuals suffering from autoimmune disorders.
Neuroinflammation
Brain-Adjacent Formulations
Neuroinflammation poses a significant challenge in treating various neurological disorders. Innovative brain-adjacent formulations are being designed to respond to inflammatory markers such as IL-1β or NF-κB activation. These formulations can release therapeutic agents before neuronal damage becomes irreversible, potentially preventing the progression of neurodegenerative diseases. By integrating real-time monitoring of inflammatory signals, these systems can provide timely interventions, thereby preserving cognitive function and improving patient outcomes.
Transplant Rejection Monitoring
Early Immune Activation Sensing
Transplant rejection remains a critical concern in organ transplantation. New implants are being developed that can sense early immune activation transcripts, triggering local immunomodulation before graft failure occurs. By identifying the molecular signals associated with rejection at an early stage, these systems can initiate targeted interventions to protect the transplanted organ. This proactive approach not only enhances graft survival rates but also reduces the need for systemic immunosuppressive therapies, which can have significant side effects.
Metabolic Disease Management
RNA-Responsive Depots
In the management of metabolic diseases, RNA-responsive depots are being explored to dynamically adjust hormone or enzyme delivery based on metabolic gene expression. These systems can monitor the levels of specific RNA transcripts associated with metabolic processes and respond accordingly by releasing the necessary therapeutic agents. This level of integration allows for a more personalized approach to managing conditions such as diabetes or obesity, as treatment can be tailored to the individual’s metabolic state in real time.
Challenges & Hidden Barriers in Advancing Technology
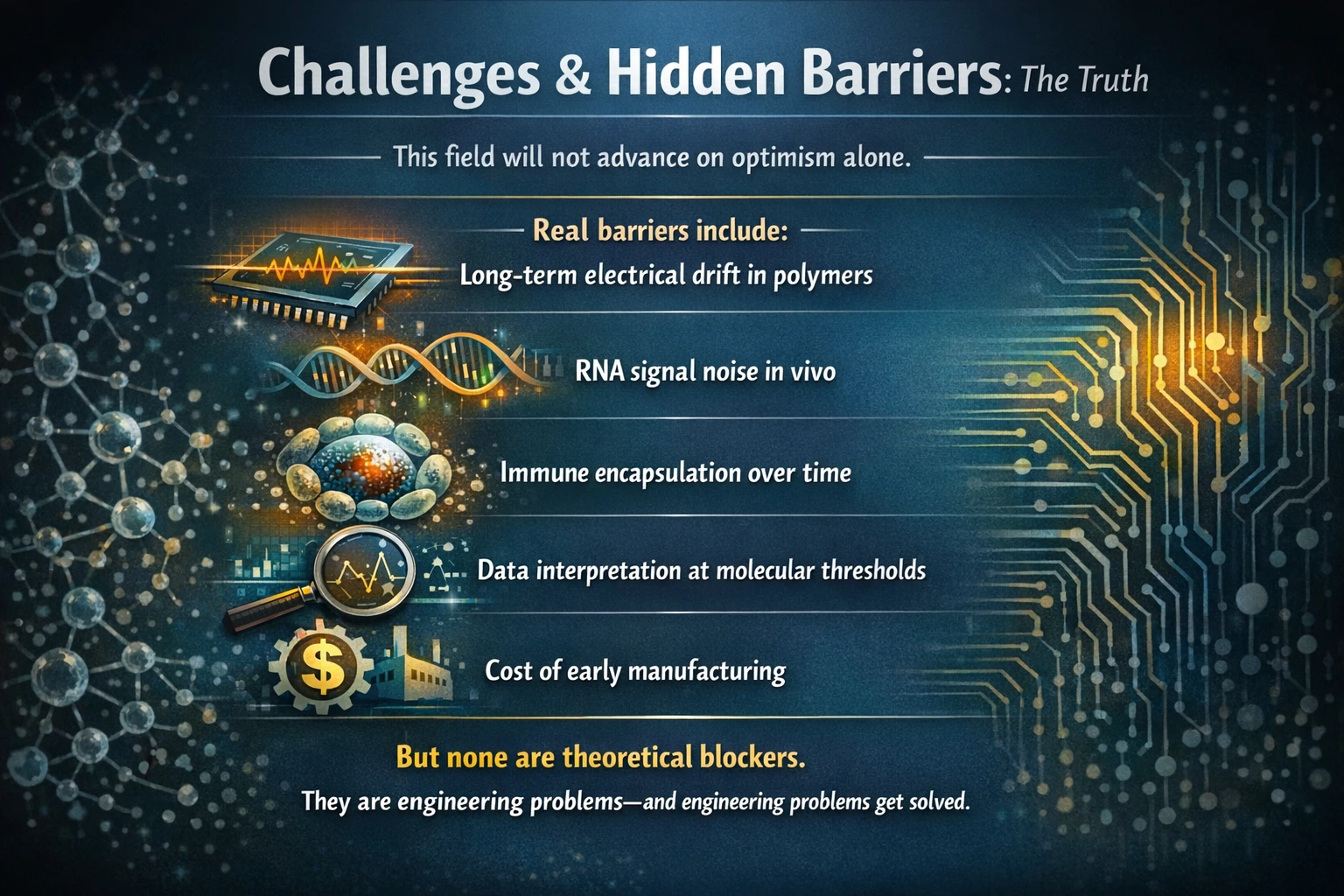
The real challenges and hidden barriers that impede progress in the field, emphasizing that optimism alone is insufficient for advancement. While these barriers are significant, they are not insurmountable; they represent engineering problems that can be addressed through innovation and persistence.
Real Barriers to Advancement
- Long-term Electrical Drift in Polymers
Polymers used in electronic applications often experience electrical drift over time, which can lead to performance degradation. This long-term instability poses a challenge for reliable device operation.
- RNA Signal Noise In Vivo
In biological systems, RNA signals can be obscured by noise, complicating the interpretation of molecular data. This variability can hinder the accuracy of biological assays and diagnostics.
- Immune Encapsulation Over Time
The body’s immune response can lead to encapsulation of foreign materials, affecting the long-term efficacy of implanted devices. This biological barrier can limit the functionality and lifespan of medical technologies.
- Data Interpretation at Molecular Thresholds
The interpretation of data at molecular levels often faces challenges due to the inherent variability and complexity of biological systems. Accurate analysis is crucial for advancing research and applications.
- Cost of Early Manufacturing
The initial costs associated with manufacturing new technologies can be prohibitively high. This financial barrier can slow down the transition from research to practical application.
The Grand Finale Conclusion: The End of Passive Medicine
For more than a century, medicine has operated on a fundamentally reactive model: detect symptoms, confirm disease, and then intervene. Even the most advanced controlled-release systems, biologics, and targeted therapies remain bound to this sequence. They improve precision, but they do not change timing. Intervention still follows manifestation.
Across this series, a different paradigm has been constructed step by step. Part 1 introduced electrogenomics as a shift from symptom-driven to signal-driven medicine. Part 2 demonstrated that this shift is materially feasible, grounded in conductive polymers, aptamer-based hydrogels, RNA-responsive nanoparticles, and magnetoelectric scaffolds. Part 3 translated these materials into a formulation workflow, showing how a scientist can move from gene-level biomarkers to a manufacturable, testable system. This final section places that system inside the human body, within clinical reality, and within the constraints of ethics, regulation, and manufacturing.
The convergence of these elements leads to a single, unavoidable conclusion: electrogenomic formulations redefine the moment at which medicine acts.
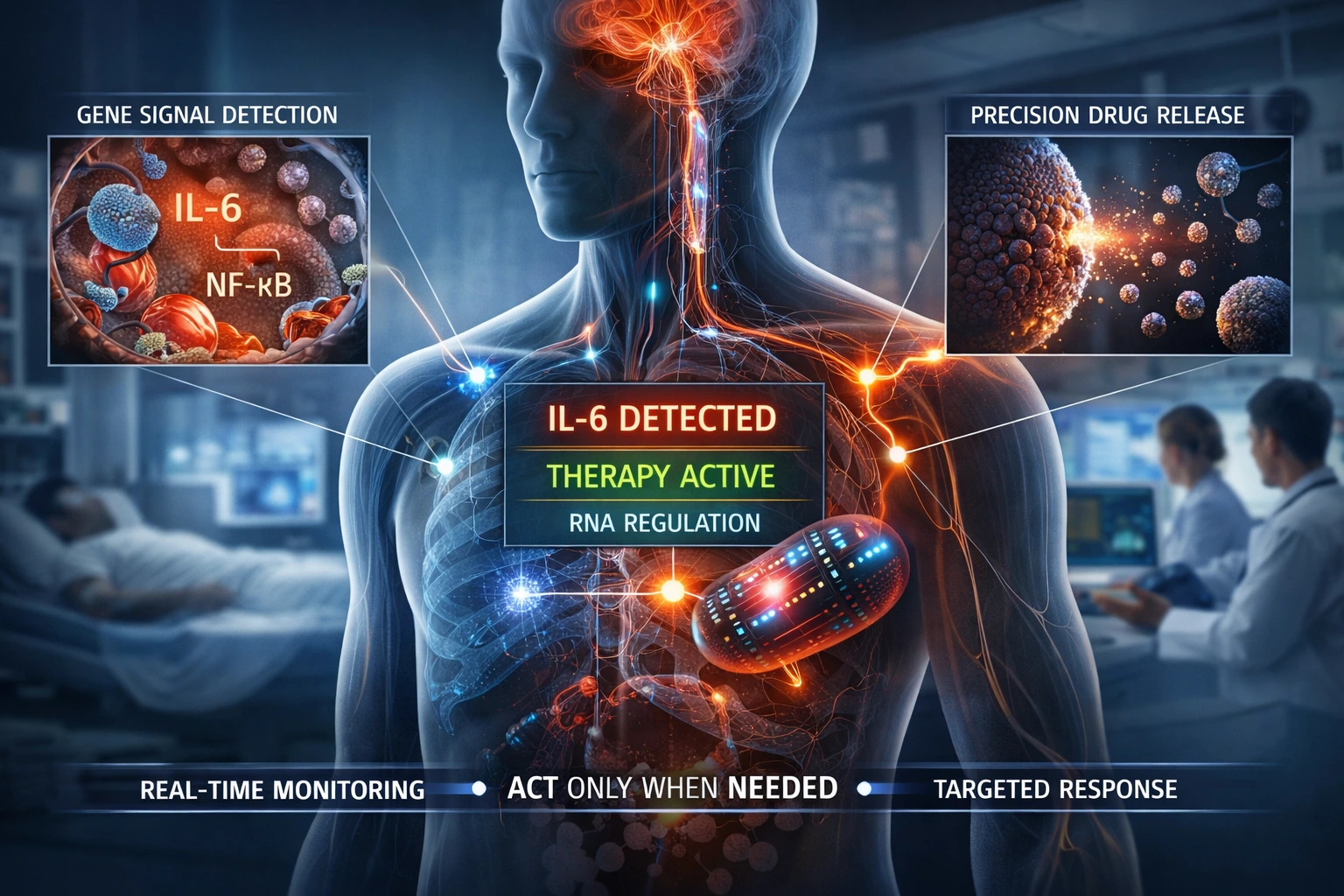
Instead of responding to downstream consequences such as inflammation, tissue damage, or clinical relapse, these systems operate at the level of gene expression itself. Molecular events such as IL-6 upregulation, NF-κB activation, or oncogenic transcript emergence are no longer passive indicators, they become actionable triggers. Through embedded sensing mechanisms, threshold logic, and controlled actuation, the formulation continuously evaluates biological state and intervenes only when predefined molecular conditions are met.
This transforms drug delivery from a time-based or concentration-based model into a conditional one. Therapy is no longer administered because a dosing schedule dictates it, but because a biological signal justifies it. Equally important, therapy is withheld when those conditions are not met. In this framework, non-action is not a limitation; it is a designed feature that prevents overtreatment, reduces systemic exposure, and preserves physiological balance.
The implications are significant. In oncology, treatment can align with tumor signaling dynamics rather than fixed cycles. In autoimmune disease, biologic release can be restricted to flare states instead of continuous suppression. In neurodegeneration and epilepsy, intervention can occur at the earliest molecular signatures rather than after irreversible damage or electrical events. Across these applications, the common advantage is not simply improved efficacy, but improved timing and contextual precision.
From a pharmaceutical perspective, this does not invalidate existing principles of formulation science, it extends them. The same rigor applied to stability, release kinetics, excipient compatibility, and scale-up now applies to signal fidelity, molecular specificity, and threshold definition. The formulation scientist is no longer optimizing only how a drug is delivered, but under what biological conditions delivery should occur.
Electrogenomics therefore does not represent an incremental improvement in drug delivery. It represents a transition from passive to responsive systems, where formulations are designed to interpret biological information in real time and act accordingly. The materials, tools, and early clinical precedents already exist. What remains is integration, validation, and the willingness to adopt a fundamentally different model of therapeutic control.
The future of medicine will not be defined solely by the molecules we develop, but by the intelligence embedded in how and when those molecules are deployed. Electrogenomic formulations mark the point at which medicine begins to operate at the same level of precision as the biology it aims to treat.
Electrogenomics defines a new class of therapy where electrogenomics replaces fixed dosing with conditional, signal-driven intervention. Through electrogenomics, formulations sense, decide, and respond—making electrogenomics the bridge between molecular biology and real-time medicine. In the end, electrogenomics is not an advancement but a shift, where electrogenomics transforms passive treatment into intelligent, responsive care, and establishes electrogenomics as the future foundation of therapeutics.
References
- Langer R., Tirrell D. Designing materials for biology and medicine. Nature
- Deans T.L., et al. Programmable biomolecular feedback systems. Cell
- Hou X., et al. Lipid nanoparticles for mRNA delivery. Nat Rev Mater
- Green A.A., et al. Toehold switches and RNA sensing circuits. Cell
- FDA. Combination Products Regulatory Guidance
- Medtronic Neuromodulation Technical Briefs
- Moderna Therapeutics Platform Publications


