Introduction to Part 3: From Materials to Method
Part 1 established the conceptual foundation of electrogenomics, introducing medicines that do not merely deliver therapy, but sense molecular states and respond accordingly. Part 2 grounded that vision in reality, detailing the material platforms that make such behavior possible: RNA-responsive nanoparticles, conductive polymers, aptamer hydrogels, and magnetoelectric scaffolds. Together, they answered two essential questions: what electrogenomics is and what it is made of.
Part 3 addresses the question that inevitably follows:
How does a formulation scientist actually build one?
This section is deliberately practical. It moves beyond describing materials and begins assembling them, step by step, into a functional electrogenomic formulations. The focus shifts from individual components to workflow, thresholds, trade-offs, and failure points: how biomarkers are chosen, how aptamers are designed, how polymers are selected for both conductivity and biocompatibility, how mRNA thresholds are defined, how stability is preserved, and how such systems are ultimately characterized, scaled, and manufactured under real pharmaceutical constraints.
Readers encountering this part for the first time are strongly encouraged to begin with Part 1 to understand the governing concept, and Part 2 to understand the material intelligence involved. Without that context, Part 3 will appear unconventional, not because it is speculative, but because it reframes formulation science around decision logic rather than dose alone.
What follows is not a hypothetical framework or a futuristic roadmap. It is a construction narrative written for formulation scientists, those trained to convert complexity into control. The tools are already familiar. The mindset is what changes.
If electrogenomic Formulations is to move from possibility to practice, this is where that transition must occur.
RNA-Responsive Nanoparticles
RNA-responsive nanoparticles represent a cutting-edge approach in targeted drug delivery, engineered to detect and react to intracellular RNA levels. These sophisticated systems, often constructed as lipid-polymer hybrids, employ various mechanisms like RNA-binding peptides, CRISPR-associated sensing elements, and strand-displacement circuits to achieve precise control over payload release. Upon reaching a predetermined threshold of mRNA, these nanoparticles undergo structural changes, releasing therapeutic agents such as siRNA, antisense oligos, or small molecules directly at the site of action. This targeted approach holds immense promise for personalized medicine and the treatment of a wide range of diseases.
RNA-responsive nanoparticles are designed to recognize intracellular RNA levels and respond accordingly. These nanoparticles are often lipid-polymer hybrids incorporating:
- RNA-binding peptides
- CRISPR-associated sensing elements
- Strand-displacement circuits
Let’s delve into each of these components and their roles in creating RNA-responsive nanoparticles:
1. RNA-binding peptides:
These peptides are specifically engineered to bind to target RNA sequences within the cell. Their incorporation into nanoparticles allows for selective recognition of cells expressing particular RNA molecules.
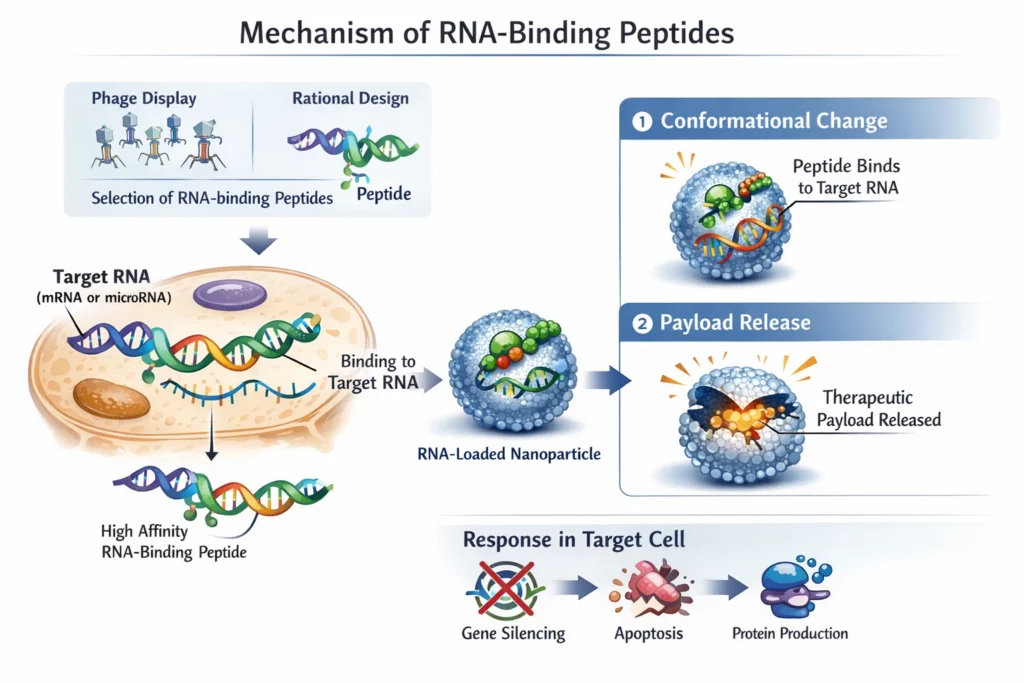
Mechanism: RNA-binding peptides are designed with amino acid sequences that exhibit high affinity and specificity for target RNA sequences, such as specific mRNA transcripts or microRNAs. These peptides can be identified through methods like phage display or rational design based on known RNA-protein interactions. When the nanoparticle encounters a cell expressing the target RNA, the RNA-binding peptide interacts with the RNA, triggering a conformational change in the nanoparticle. This change can lead to the release of the therapeutic payload or initiate other desired responses.
- Advantages: High specificity for target RNA sequences, potential for modular design and easy incorporation into nanoparticles.
- Limitations: Potential for off-target binding, immunogenicity, and limited ability to penetrate cell membranes without proper modification.
2. CRISPR-associated sensing elements:
These elements leverage the specificity of CRISPR-Cas systems to detect and respond to specific RNA sequences.
Mechanism: CRISPR-associated sensing elements utilize the Cas protein’s ability to bind to a guide RNA (gRNA) that is complementary to a target RNA sequence. When the target RNA is present, the Cas protein binds to it, triggering a downstream event. In the context of RNA-responsive nanoparticles, this event can be the activation of a payload release mechanism. For example, the Cas protein could be engineered to cleave a linker molecule that holds the therapeutic payload within the nanoparticle, leading to its release. Alternatively, the binding of the Cas protein to the target RNA could induce a conformational change in the nanoparticle, exposing a previously hidden therapeutic agent.
- Advantages: Highly specific and programmable RNA detection, potential for multiplexed RNA sensing by incorporating multiple gRNAs.
- Limitations: Potential for off-target effects, immunogenicity of Cas proteins, and challenges in delivering large CRISPR-Cas complexes into cells.
3. Strand-displacement circuits:
These circuits utilize the principles of nucleic acid hybridization to create dynamic and responsive systems that can detect and respond to specific RNA sequences.
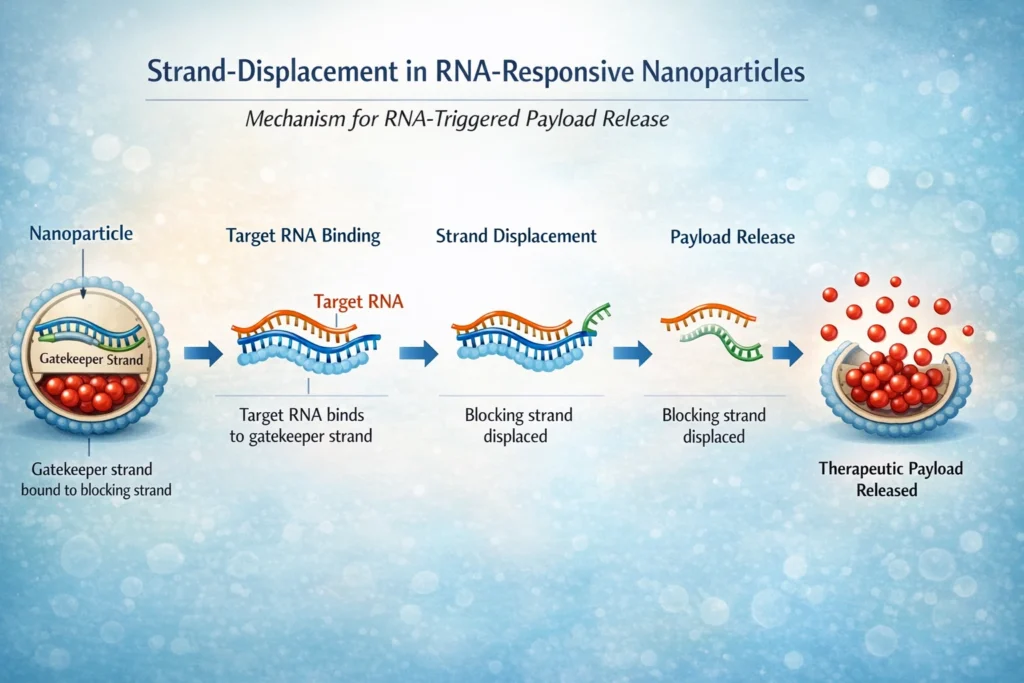
Mechanism: Strand-displacement circuits are based on the competition between different nucleic acid strands for binding to a target sequence. In the context of RNA-responsive nanoparticles, these circuits can be designed to release a therapeutic payload when a target RNA is present. For example, the nanoparticle could contain a “gatekeeper” strand that is hybridized to a complementary strand, preventing the release of the payload. When the target RNA is present, it can bind to the gatekeeper strand with higher affinity, displacing the complementary strand and opening the gate for payload release.
- Advantages: Highly tunable and programmable, potential for complex logic operations, and relatively simple design.
- Limitations: Potential for non-specific interactions, sensitivity to environmental conditions, and limited stability in biological environments.
Upon detection of threshold mRNA levels, these nanoparticles undergo structural rearrangement, releasing payloads such as siRNA, antisense oligos, or small molecules.
- siRNA (small interfering RNA): siRNA molecules are short, double-stranded RNA molecules that can silence gene expression by targeting specific mRNA transcripts for degradation.
- Antisense oligos (antisense oligonucleotides): Antisense oligos are short, single-stranded DNA or RNA molecules that can bind to specific mRNA transcripts and inhibit their translation into proteins.
- Small molecules: Small molecules are low-molecular-weight organic compounds that can exert a variety of therapeutic effects by interacting with specific biological targets.
Clinical trajectory: Moderna and BioNTech are exploring RNA-sensing lipid nanoparticles that adjust mRNA translation efficiency based on intracellular inflammatory markers. This approach has the potential to improve the efficacy and safety of mRNA-based therapies by tailoring the dose and duration of treatment to the individual patient’s needs. By monitoring inflammatory markers, these nanoparticles can dynamically adjust the amount of mRNA translated into protein, ensuring that the therapeutic effect is optimized while minimizing the risk of adverse events. This represents a significant step towards personalized medicine and the development of more effective and targeted therapies.
Magnetoelectric Scaffolds
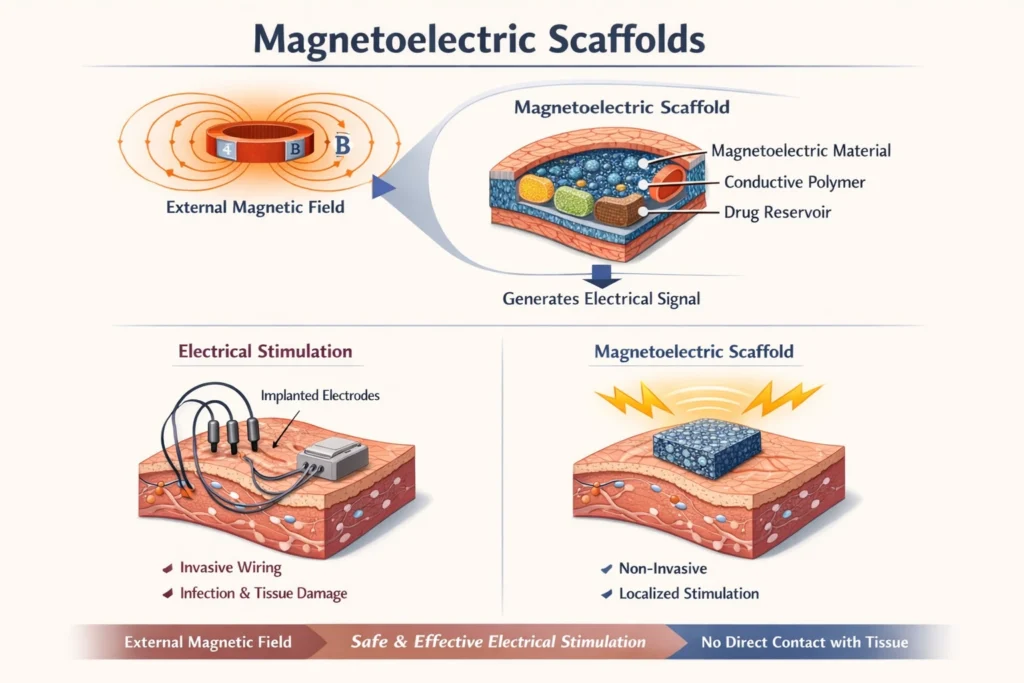
Magnetoelectric materials possess the unique ability to convert magnetic fields into electrical signals. This property makes them highly valuable in various applications, particularly in the realm of biomedical engineering. When these materials are integrated into polymer scaffolds, they create a platform for wirelessly controlling electrogenomic Formulations systems.
The core principle behind this technology lies in the interaction between an external magnetic field and the magnetoelectric material. When exposed to a magnetic field, the material generates an electrical potential. This electrical signal can then be harnessed to activate various components within the scaffold, such as conductive polymers or drug reservoirs.
One of the key advantages of magnetoelectric scaffolds is their ability to provide localized electrical stimulation without the need for invasive wiring. Traditional methods of electrical stimulation often require the implantation of electrodes, which can lead to complications such as infection, inflammation, and tissue damage. Magnetoelectric scaffolds offer a non-invasive alternative, as the magnetic field can be applied externally, eliminating the need for direct contact with the target tissue.
This non-invasive nature makes magnetoelectric scaffolds particularly attractive for applications in regenerative medicine. For example, they can be used to stimulate nerve regeneration after spinal cord injury. By embedding magnetoelectric composites in a scaffold and implanting it at the site of injury, external magnetic fields can be used to generate electrical signals that promote nerve growth and functional recovery.
The electrical signals generated by the magnetoelectric material can also be used to activate conductive polymers within the scaffold. Conductive polymers are materials that can conduct electricity when stimulated. By incorporating these polymers into the scaffold, it is possible to create a system that can deliver electrical stimulation to specific cells or tissues in a controlled manner. This can be useful for a variety of applications, such as promoting wound healing, stimulating bone growth, or modulating gene expression.
Another potential application of magnetoelectric scaffolds is in the controlled release of drugs. By incorporating drug reservoirs into the scaffold and using the electrical signal generated by the magnetoelectric material to open these reservoirs, it is possible to deliver drugs to specific locations in the body on demand. This can be particularly useful for treating conditions such as cancer, where targeted drug delivery is essential to minimize side effects.
Emerging Case: Spinal Cord Implants
One of the most promising applications of magnetoelectric scaffolds is in the development of spinal cord implants. Spinal cord injury (SCI) is a devastating condition that can lead to permanent paralysis and loss of function. While there is currently no cure for SCI, researchers are exploring various strategies to promote nerve regeneration and functional recovery.
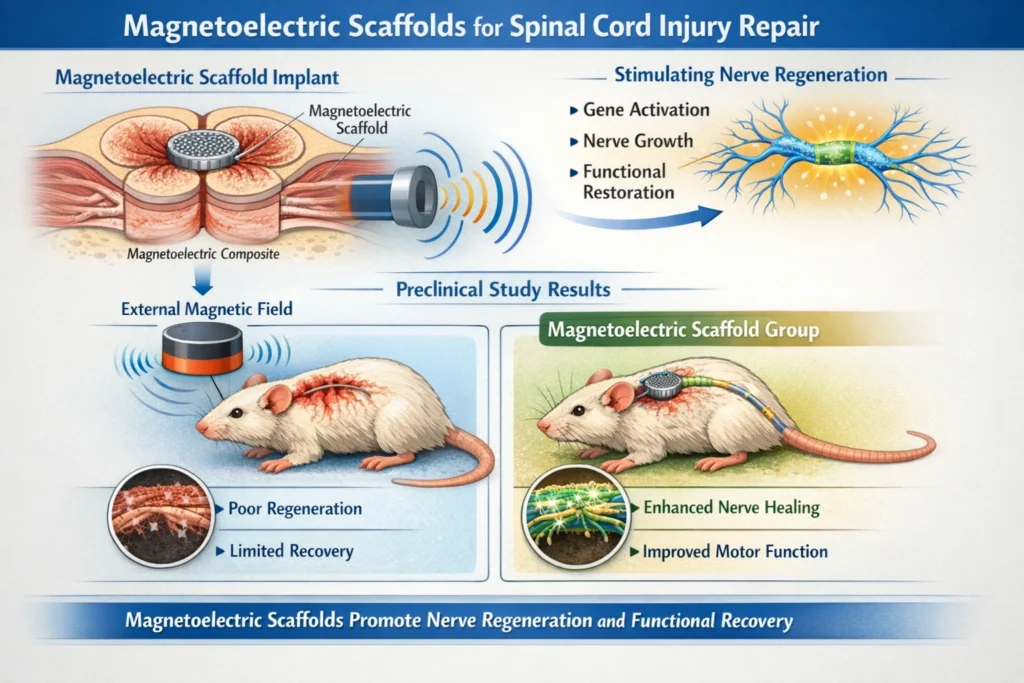
Magnetoelectric scaffolds offer a potential solution for stimulating nerve regeneration after SCI. By embedding magnetoelectric composites in a scaffold and implanting it at the site of injury, external magnetic fields can be used to generate electrical signals that promote nerve growth. These electrical signals can activate genes related to neural regeneration, leading to the formation of new nerve connections and the restoration of function.
Experimental spinal cord implants using magnetoelectric composites have shown promising results in preclinical studies. In these studies, animals with SCI were treated with magnetoelectric scaffolds, and their recovery was compared to that of control animals. The results showed that the animals treated with magnetoelectric scaffolds had significantly better nerve regeneration and functional recovery than the control animals.
These findings suggest that magnetoelectric scaffolds have the potential to revolutionize the treatment of SCI. However, further research is needed to optimize the design and performance of these scaffolds and to evaluate their safety and efficacy in human clinical trials.
Challenges and Future Directions
While magnetoelectric scaffolds hold great promise for various biomedical applications, there are also several challenges that need to be addressed. One of the main challenges is the development of magnetoelectric materials with high efficiency and biocompatibility. The ideal magnetoelectric material should be able to generate a strong electrical signal in response to a weak magnetic field, and it should be non-toxic and well-tolerated by the body.
Another challenge is the design of scaffolds that can effectively deliver the electrical signal to the target tissue. The scaffold should be porous enough to allow for cell infiltration and tissue ingrowth, but it should also be strong enough to withstand the mechanical forces of the body. Additionally, the scaffold should be designed to ensure that the electrical signal is delivered to the specific cells or tissues that need to be stimulated.
Future research in this area will focus on developing new magnetoelectric materials with improved properties, designing scaffolds with optimized architectures, and conducting preclinical and clinical studies to evaluate the safety and efficacy of magnetoelectric scaffolds in various applications. With continued research and development, magnetoelectric scaffolds have the potential to transform the treatment of a wide range of diseases and injuries.
How a Formulator Would Actually Build One (Step-by-Step)
The practical steps a formulator would take to build an electrogenomic Formulations system, moving from theoretical concepts to tangible lab procedures. It details the critical considerations at each stage, from biomarker selection to scale-up, emphasizing the unique challenges and requirements of this emerging field.
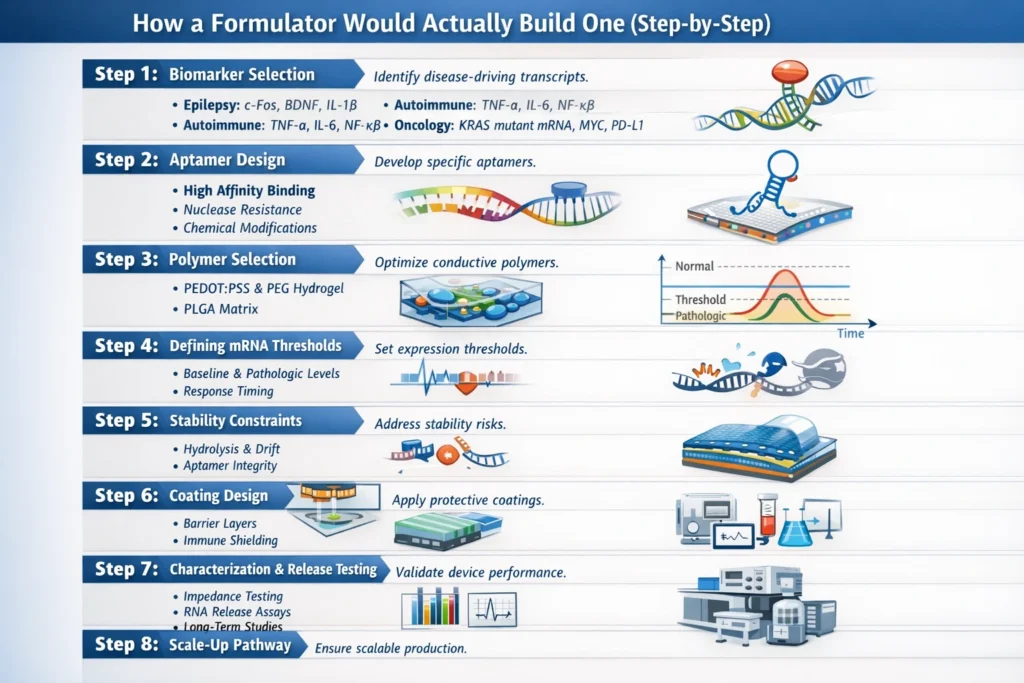
Step 1: Biomarker Selection
The initial step in electrogenomic formulation involves identifying specific disease-driving transcripts, shifting the focus from treating symptoms to directly addressing the underlying molecular causes.
Examples:
- Epilepsy: c-Fos, BDNF, IL-1β
- Autoimmune disease: TNF-α, IL-6, NF-κB
- Oncology: KRAS mutant mRNA, MYC, PD-L1
Selection Criteria:
- Expression Amplitude: The transcript should exhibit a significant change in expression levels during disease states.
- Temporal Dynamics: The timing of transcript expression relative to disease progression is crucial for designing responsive systems.
- Tissue Specificity: Targeting transcripts predominantly expressed in affected tissues minimizes off-target effects.
Step 2: Aptamer Design
Aptamers, short single-stranded DNA or RNA molecules, are engineered to selectively bind to the chosen mRNA transcripts.
Design Considerations:
- Binding Affinity (Kd in low nM range): High affinity ensures efficient target capture.
- Specificity against Homologous Sequences: Aptamers must discriminate against similar transcripts to avoid unintended interactions.
- Stability against Nucleases: Aptamers are susceptible to degradation by nucleases in biological environments.
Chemical Modifications:
- 2’-O-methyl: Enhances nuclease resistance and improves binding affinity.
- Phosphorothioate: Replaces a non-bridging oxygen in the phosphate backbone with sulfur, increasing nuclease resistance.
These modifications extend the in vivo half-life of the aptamer, improving its therapeutic efficacy.
Step 3: Polymer Selection
The polymer matrix serves as the structural foundation of the electrogenomic Formulations system, providing a scaffold for aptamers and enabling electrical conductivity.
Key Requirements:
- Electrical Conductivity: The polymer must facilitate electron transport to enable transcript detection and therapeutic response.
- Mechanical Compliance: The matrix should be flexible and conformable to the target tissue to minimize irritation and ensure biocompatibility.
- Biocompatibility: The polymer must be non-toxic and elicit minimal immune response.
Common Combinations:
- PEDOT:PSS blended with PEGylated hydrogels: PEDOT:PSS provides electrical conductivity, while PEGylated hydrogels offer biocompatibility and mechanical flexibility.
- PLGA-based matrices: PLGA (poly(lactic-co-glycolic acid)) is a biodegradable polymer that can be formulated into porous matrices for controlled release and tissue integration.
Step 4: Defining mRNA Thresholds
Electrogenomic Formulations systems operate on a threshold-based principle, responding to specific levels of target transcripts rather than simply detecting their presence or absence.
Formulation Steps:
- Baseline Transcript Levels: Determine the normal expression levels of the target transcript in healthy tissue.
- Pathological Overexpression Thresholds: Identify the transcript levels that indicate disease onset or progression.
- Response Latency Windows: Define the time frame within which the system should respond to changes in transcript levels.
Data Acquisition:
- Transcriptomic Datasets: Analyze existing gene expression data to establish baseline and pathological transcript levels.
- In vitro Cell Models: Use cell cultures to validate threshold levels and optimize system response.
Step 5: Stability Constraints
Maintaining the stability of the electrogenomic Formulations system is critical for its long-term performance and efficacy.
Key Risks:
- Hydrolytic Degradation: Polymers and aptamers can degrade in the presence of water.
- Electrical Drift: The electrical properties of the system can change over time, affecting its sensitivity and accuracy.
- Aptamer Denaturation: Aptamers can lose their structure and binding affinity under certain conditions.
Stability Protocols:
- Accelerated Stability Testing: Expose the system to elevated temperatures and humidity levels to simulate long-term storage conditions.
- Implantable Device Guidelines: Adapt stability protocols from the implantable device industry, which has extensive experience in ensuring the long-term performance of medical devices.
Step 5: Stability Constraints
Maintaining the stability of the electrogenomic Formulations system is critical for its long-term performance and efficacy.
Key Risks:
- Hydrolytic Degradation: Polymers and aptamers can degrade in the presence of water.
- Electrical Drift: The electrical properties of the system can change over time, affecting its sensitivity and accuracy.
- Aptamer Denaturation: Aptamers can lose their structure and binding affinity under certain conditions.
Stability Protocols:
- Accelerated Stability Testing: Expose the system to elevated temperatures and humidity levels to simulate long-term storage conditions.
- Implantable Device Guidelines: Adapt stability protocols from the implantable device industry, which has extensive experience in ensuring the long-term performance of medical devices.
Step 6: Coating Design
Protective coatings are applied to the electrogenomic Formulations system to regulate its interaction with the surrounding environment.
Coating Functions:
- Water Ingress: Control the rate at which water penetrates the system to prevent degradation.
- Ion Diffusion: Regulate the movement of ions into and out of the system to maintain electrical conductivity.
- Immune Recognition: Minimize immune response by masking the system from immune cells.
Coating Materials:
- Layer-by-Layer Polyelectrolyte Coatings: Alternating layers of positively and negatively charged polymers create a stable and biocompatible barrier.
Step 7: Characterization & Release Testing
Thorough characterization and release testing are essential to ensure the quality and performance of the electrogenomic Formulations system.
Testing Methods:
- Electrical Impedance Spectroscopy: Measures the electrical properties of the system to assess its conductivity and stability.
- Transcript-Triggered Release Assays: Evaluates the system’s ability to release therapeutic agents in response to target transcripts.
- Long-Term Signal Fidelity Studies: Monitors the system’s ability to maintain a stable and accurate signal over time.
Step 8: Scale-Up Pathway
Scaling up the manufacturing process is crucial for translating electrogenomic Formulations systems from the lab to clinical applications.
Scale-Up Considerations:
- Microfabrication Reproducibility: Ensure that the microfabrication process can be reliably replicated at larger scales.
- Polymer Batch Consistency: Maintain consistent quality and properties of the polymer materials used in the system.
- Sterilization Compatibility: Develop sterilization methods that do not compromise the system’s performance.
Manufacturing Alignment:
Combination Products: Electrogenomic Formulations system are more akin to combination products (drug-device combinations) than classical pharmaceuticals, requiring expertise in both drug formulation and device manufacturing.
Conclusion: When Formulation Becomes a Living Process
Magnetoelectric scaffolds extend this logic beyond chemistry into physics. By enabling wireless, localized activation, they dissolve the historical boundary between implantable devices and drug delivery systems. Here, formulation is no longer sealed at administration, it remains addressable, modulatable, and responsive long after placement.
Part 3 brings electrogenomic Formulations to its most consequential point, not as a theory, not as a collection of smart materials, but as a buildable, testable pharmaceutical reality. RNA-responsive nanoparticles and magnetoelectric scaffolds demonstrate that modern formulations are no longer constrained to passive delivery. They can sense intracellular states, translate molecular signals into physical action, and intervene only when biology demands it.
RNA-responsive nanoparticles represent a decisive shift in delivery logic. Instead of forcing expression or silencing indiscriminately, they wait, monitoring intracellular mRNA thresholds and responding with structural precision. In this framework, therapy becomes conditional, proportional, and reversible. The formulation does not dominate the cell; it negotiates with it.
The step-by-step workflow presented in this part reframes Electrogenomic formulations science itself. Biomarker selection replaces dose selection as the first design decision. Aptamers act as molecular selectors. Polymers become logic carriers. Stability is defined not only by chemical integrity, but by signal fidelity. Scale-up resembles combination-product manufacturing rather than classical solid dosage production.
What ultimately emerges is a new identity for the formulation scientist, not merely a developer of dosage forms, but an architect of molecular decision systems. The materials already exist. The biological signals are well characterized. The regulatory pathways are beginning to converge.
Electrogenomic formulations do not promise louder medicine or stronger medicine, but quieter, earlier, and more precise intervention. They act only when disease declares itself at the molecular level, and they remain silent when it does not.
At this point, the question is no longer whether such systems are feasible. It is whether formulation science is ready to accept that the medicine is now listening.
References
- Paunovska K, Loughrey D, Dahlman JE. Drug delivery systems for RNA therapeutics. Nature Reviews Genetics, 2022.
- Kulkarni JA et al. Lipid nanoparticle technology for clinical translation of siRNA and mRNA therapeutics. Accounts of Chemical Research, 2019.
- Chen Y, Ke G, et al. DNA/RNA strand-displacement circuits for biosensing and controlled drug release. Chemical Reviews, 2020.
- Hou X et al. Lipid nanoparticles for mRNA delivery. Nature Reviews Materials, 2021.
- Martins P et al. Magnetoelectric materials for biomedical applications. Advanced Functional Materials, 2018.
- Li P et al. Implantable magnetoelectric bioelectronics for wireless stimulation and therapy. Biosensors and Bioelectronics, 2023.
- Ribeiro C, Sencadas V, et al. Piezoelectric and magnetoelectric polymers for tissue engineering. Progress in Polymer S Nezakati T et al. Conductive polymers in biomedical engineering. Chemical Reviews, 2018.
- Sefah K et al. DNA aptamers and their biological applications. Chemical Reviews, 2019.
- Svirskis D et al. Electrochemically controlled drug delivery using conducting polymers. Journal of Controlled Release, 2010.
- FDA. Guidance on Combination Products and Human Factors Engineering.
- ICH Q8–Q12 (Pharmaceutical Development, Quality Risk Management, Lifecycle Management).
