Introduction: R&D and F&D Concept
In the pharmaceutical world, Research & Development (R&D) is often celebrated as the backbone of innovation. It’s where molecules are discovered, mechanisms are decoded, and therapies are envisioned. However, behind every successful product lies another equally critical, often underestimated domain, Formulation & Development (F&D). While R&D focuses on identifying potential therapeutic agents, F&D is responsible for transforming these discoveries into viable, effective, and safe medications. If R&D discovers the “what,” F&D determines the “how.” Without the “how,” the “what” rarely reaches the patient. This article delves into the essential role of F&D in the pharmaceutical landscape, highlighting its significance in ensuring that innovative therapies are not only developed but also delivered effectively to those in need.

In the pharmaceutical world, Research & Development (R&D) is often celebrated as the backbone of innovation. It’s where molecules are discovered, mechanisms are decoded, and therapies are envisioned. But behind every successful product lies another equally critical, often underestimated domain, Formulation & Development (F&D). If R&D discovers the what, F&D determines the how. And without the how, the what rarely reaches the patient.
Formulation & Development is not merely a step in the drug development process; it is a complex interplay of science, engineering, and regulatory considerations that transforms a promising compound into a viable therapeutic product. This article delves into the essential functions of F&D, highlighting its significance in ensuring that innovative therapies are not only effective but also safe, stable, and accessible to patients. By exploring the intricate processes involved in F&D, we aim to shed light on its pivotal role in the journey from laboratory bench to bedside, emphasizing that the success of pharmaceutical innovation hinges on both the discovery of new drugs and the meticulous development of their formulations.
Understanding the Divide, and the Connection
Research & Development (R & D)
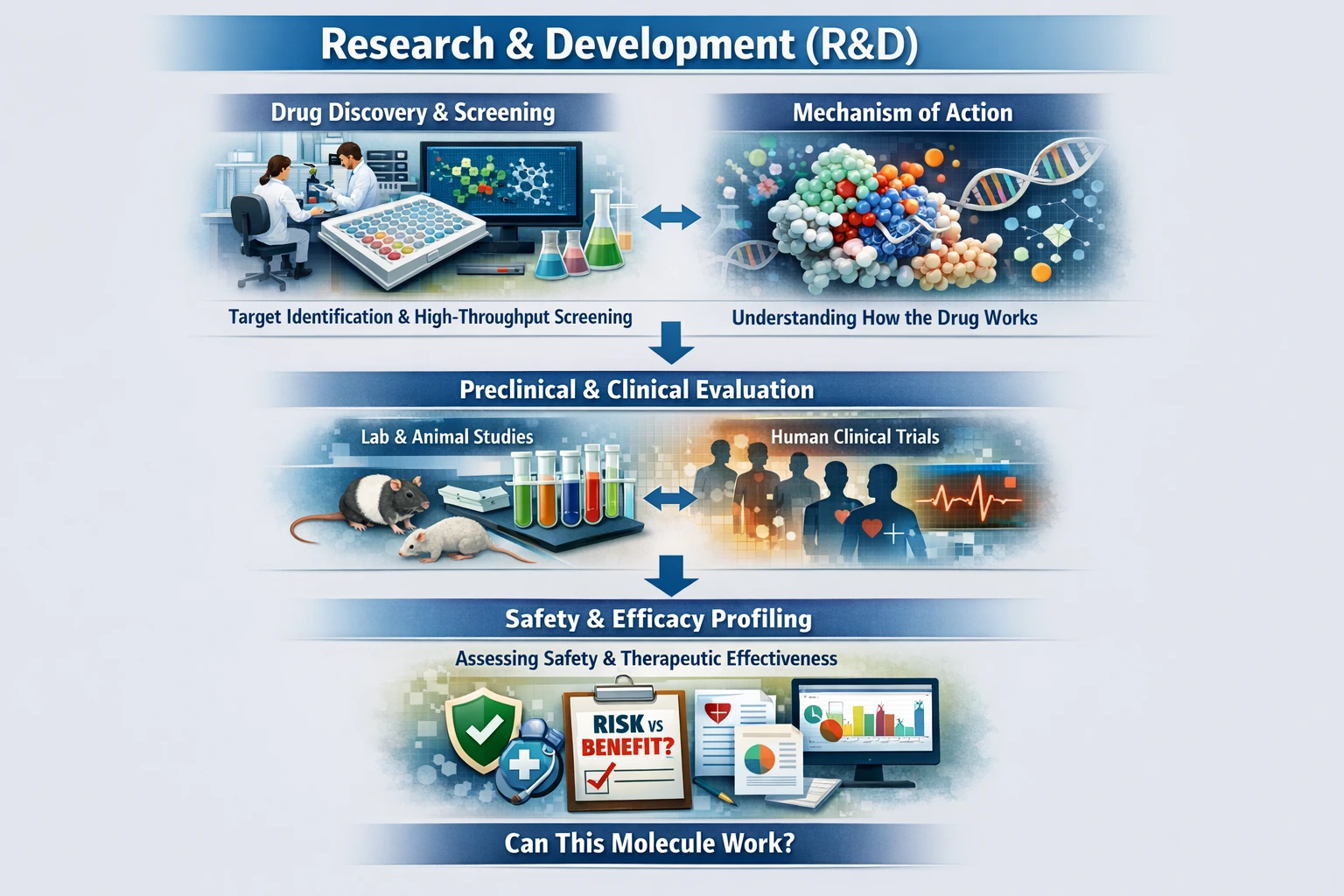
The critical phases of drug development, specifically focusing on Research & Development (R&D) and Formulation & Development (F&D). It highlights the distinct yet interconnected roles these phases play in bringing a new drug from concept to market. By examining the processes involved in drug discovery, evaluation, and formulation, we can better understand how effective medicines are created and the challenges faced along the way.
Drug Discovery and Screening
The R&D phase begins with drug discovery, where scientists identify potential drug candidates through various methods, including high-throughput screening and computational modeling. This initial stage is crucial as it lays the groundwork for subsequent research. Researchers aim to find molecules that exhibit desired biological activity against specific targets, such as proteins associated with diseases.
Mechanism of Action
Once potential candidates are identified, understanding their mechanism of action becomes essential. This involves studying how the drug interacts with biological systems at the molecular level. Knowing how a drug works helps researchers predict its effects, potential side effects, and overall therapeutic efficacy. This knowledge is vital for optimizing the drug’s design and ensuring it targets the intended pathways effectively.
Preclinical and Clinical Evaluation
After establishing a drug’s mechanism of action, the next step is preclinical evaluation, which typically involves laboratory and animal studies. These studies assess the drug’s safety, pharmacokinetics, and pharmacodynamics. If preclinical results are promising, the drug advances to clinical trials, which are conducted in phases to evaluate safety and efficacy in humans.
Safety and Efficacy Profiling
Throughout the R&D phase, safety and efficacy profiling is paramount. Researchers must ensure that the drug not only works as intended but also does not pose unacceptable risks to patients. This involves rigorous testing and data analysis to establish a favorable risk-benefit profile. Ultimately, this phase answers the critical question: Can this molecule work?
Formulation & Development (F & D)
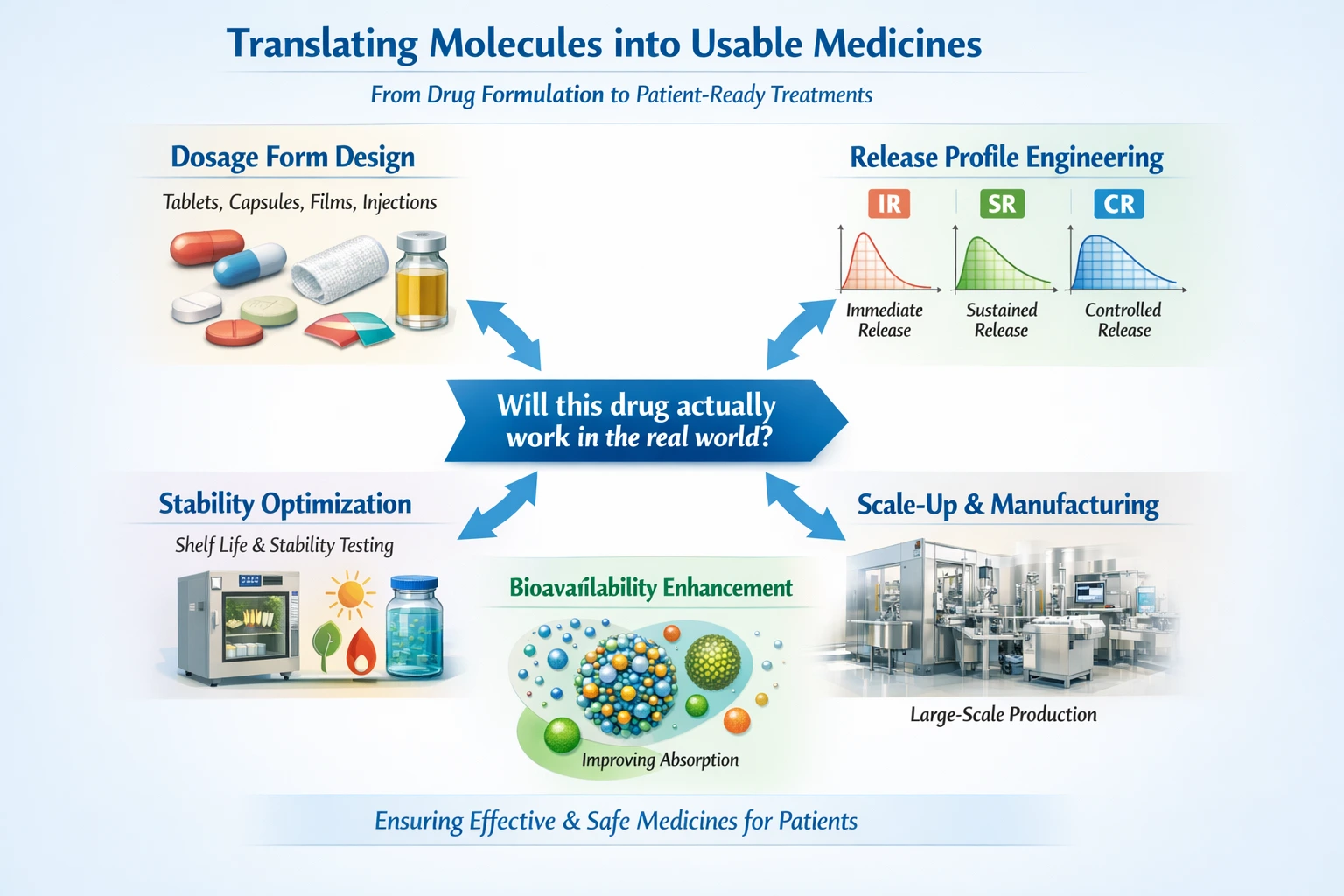
Translating Molecules into Usable Medicines
Once a drug candidate has successfully navigated the R&D phase, it enters the F&D phase, where the focus shifts to translating the molecule into a usable medicine. This involves several key processes that ensure the drug can be effectively administered to patients.
Dosage Form Design
The first step in F&D is designing the dosage form, which can take various forms such as tablets, capsules, films, or injections. The choice of dosage form is influenced by factors like the drug’s properties, the target patient population, and the intended route of administration. Each form has its advantages and challenges, and selecting the right one is crucial for patient compliance and therapeutic effectiveness.
Release Profile Engineering
Next, researchers engineer the drug’s release profile, determining how quickly and effectively the drug will be released into the body. This can involve creating immediate-release (IR), sustained-release (SR), controlled-release (CR), or targeted delivery systems. The goal is to optimize the drug’s absorption and therapeutic effect while minimizing side effects.
Stability Optimization
Stability is another critical consideration in F&D. Researchers must ensure that the drug remains effective and safe throughout its shelf life. This involves conducting stability studies under various environmental conditions to determine how factors like temperature, humidity, and light affect the drug’s integrity.
Scale-Up and Manufacturability
Once the formulation is optimized, the next challenge is scale-up and manufacturability. This phase involves developing processes that can produce the drug at a commercial scale while maintaining quality and consistency. It requires collaboration between scientists, engineers, and manufacturing experts to ensure that the drug can be produced efficiently and cost-effectively.
Bioavailability Enhancement
Finally, enhancing bioavailability is essential for ensuring that the drug reaches its target site in the body at the right concentration. This may involve modifying the formulation to improve solubility or absorption. Techniques such as nanoparticle formulation or the use of excipients can be employed to enhance bioavailability.
Answering the Critical Question
The F&D phase ultimately answers the question: Will this drug actually work in the real world? By addressing formulation challenges and ensuring that the drug can be effectively delivered to patients, F&D plays a crucial role in the success of a new medicine.
Why F&D Is Not Just “Support”, It’s Strategy!
In the world of pharmaceuticals, formulation and development (F&D) is often viewed as a mere support function. However, this perspective undermines the critical role F&D plays in transforming theoretical drug candidates into viable therapeutic products. This document explores the multifaceted challenges that can lead to the failure of promising molecules and how strategic F&D interventions can resolve these issues, ensuring that drug candidates not only survive but thrive in the competitive landscape of drug development.
Challenges Leading to Drug Candidate Failure
Even a molecule with excellent pharmacological properties can falter during the development process due to several key challenges:
Poor Solubility
Many drug candidates suffer from poor solubility, which can severely limit their bioavailability. This is particularly true for hydrophobic compounds that do not dissolve well in biological fluids, making it difficult for the body to absorb the drug effectively. Poor solubility can lead to suboptimal therapeutic outcomes, rendering even the most promising compounds ineffective.
Instability
Instability is another significant hurdle that can derail drug development. Factors such as moisture, oxidation, and light sensitivity can compromise the integrity of a drug formulation. For instance, some compounds may degrade when exposed to humidity or light, leading to reduced efficacy or even harmful byproducts. Ensuring the stability of a drug throughout its shelf life is crucial for maintaining its therapeutic potential.
Low Bioavailability
Low bioavailability is often a consequence of poor solubility and instability. Even if a drug is absorbed, it may not reach the systemic circulation in sufficient quantities to exert its intended effect. This can be particularly problematic for drugs that require a specific concentration in the bloodstream to be effective. Low bioavailability can lead to increased dosing requirements, which may not only be impractical but can also elevate the risk of side effects.
Inconsistent Release Profiles
Inconsistent release profiles can further complicate drug development. A drug that does not release its active ingredient at a predictable rate can lead to fluctuations in therapeutic levels, resulting in subtherapeutic or toxic effects. Achieving a consistent release profile is essential for ensuring that patients receive the intended dosage over time, thereby maximizing therapeutic efficacy while minimizing adverse effects.
How F&D Resolves These Challenges
Formulation and development (F&D) employs a range of strategies to address these challenges, transforming theoretical drug candidates into practical solutions:
Polymer Engineering
One of the key strategies in F&D is polymer engineering, which involves the design of controlled release matrices. By selecting appropriate polymers, formulators can create systems that release the drug at a predetermined rate, thereby improving bioavailability and ensuring consistent therapeutic levels. Controlled release formulations can also enhance patient compliance by reducing the frequency of dosing.
Coating Technologies
Coating technologies play a vital role in protecting drug formulations from environmental factors. Enteric coatings can prevent degradation in the acidic environment of the stomach, allowing the drug to be released in the more neutral pH of the intestines. Functional coatings can enhance solubility, while protective coatings can shield sensitive compounds from moisture and light. These technologies are essential for maintaining the stability and efficacy of drug products.
Microenvironmental pH Modification
Microenvironmental pH modification is another innovative approach used in F&D. By altering the pH of the drug formulation, formulators can enhance solubility and stability. This technique can be particularly useful for drugs that are pH-sensitive, allowing for improved absorption and bioavailability.
Particle Engineering and Granulation Science
Particle engineering and granulation science are critical components of F&D that focus on optimizing the physical properties of drug particles. By controlling particle size and distribution, formulators can enhance solubility and improve flow characteristics, which are essential for consistent release profiles. Granulation techniques can also improve the compressibility of powders, facilitating the production of tablets and other solid dosage forms.
In Reality: F&D as a Strategic Partner
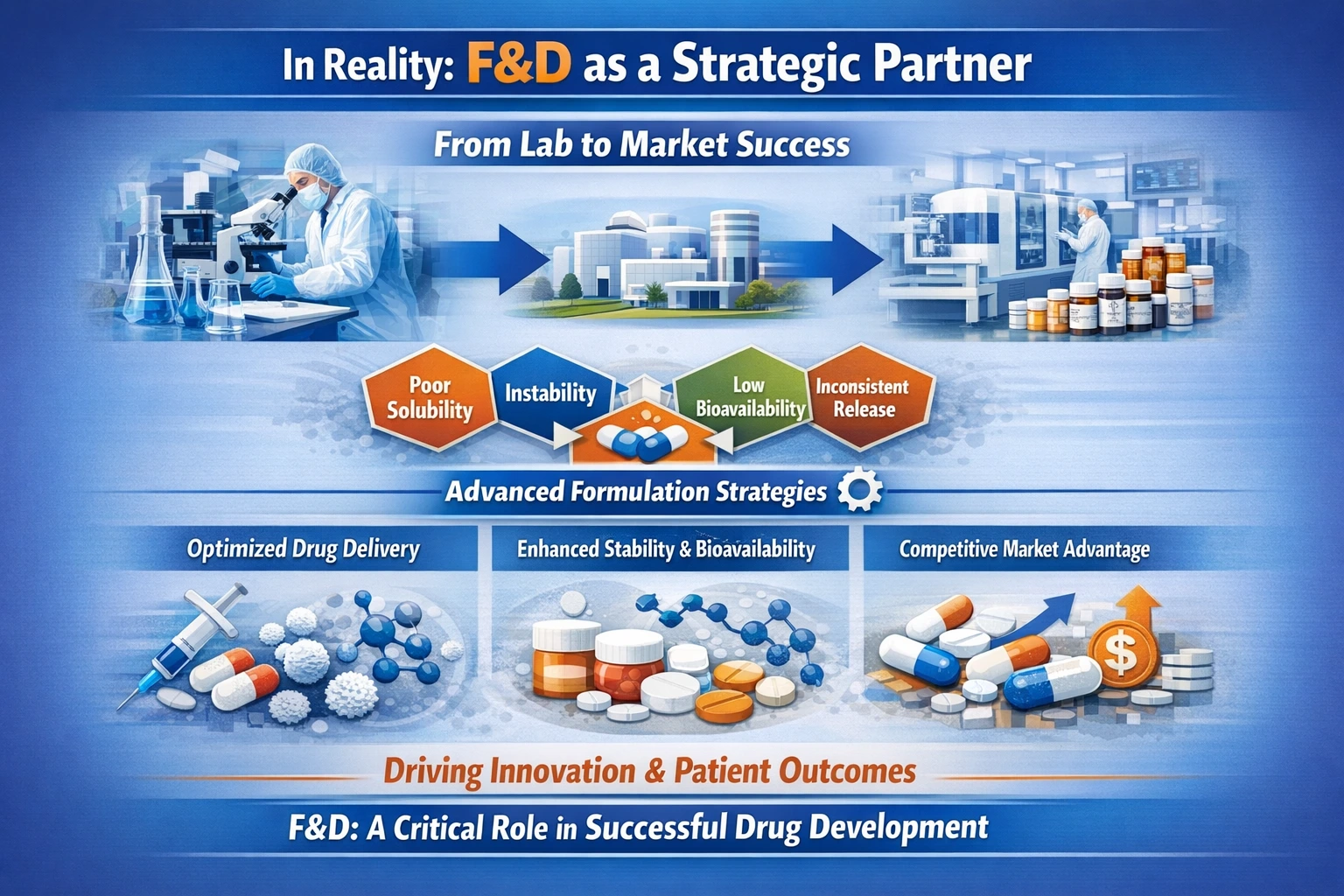
In reality, F&D is where theoretical success meets practical reality. It is not merely a support function; it is a strategic partner in the drug development process. By addressing the challenges of poor solubility, instability, low bioavailability, and inconsistent release profiles, F&D enables the successful translation of drug candidates from the lab to the market.
The integration of advanced formulation strategies ensures that drug candidates are not only viable but also competitive in a crowded marketplace. As the pharmaceutical landscape continues to evolve, the importance of F&D as a strategic function will only grow, underscoring its role in driving innovation and improving patient outcomes.
In conclusion, recognizing F&D as a strategic component of drug development is essential for fostering a culture of innovation and ensuring the success of new therapeutic agents. By investing in robust formulation strategies, pharmaceutical companies can navigate the complexities of drug development and bring effective treatments to patients in need.
In the realm of pharmaceutical development, formulation and development (F&D) is often viewed as a secondary function, a mere support system for the more glamorous aspects of drug discovery. However, this perspective overlooks the critical role that F&D plays in transforming theoretical pharmacological success into practical, market-ready products. This document explores the multifaceted challenges that can hinder drug development, even when a molecule exhibits excellent pharmacological properties, and how F&D strategies can effectively address these issues.
A Molecule with Excellent Pharmacology Can Still Fail Because of:
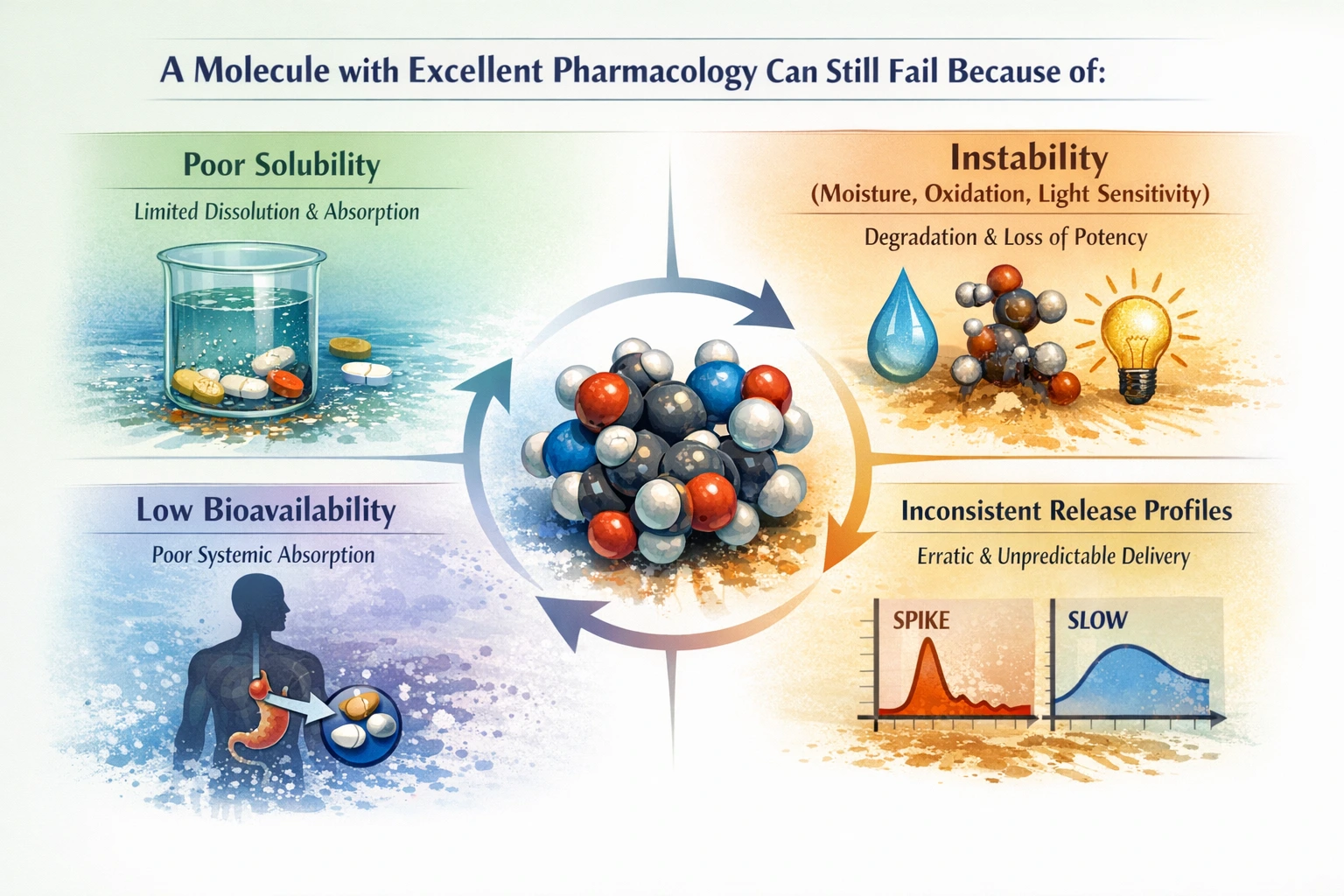
Poor Solubility
One of the most significant barriers to drug efficacy is poor solubility. Many promising compounds struggle to dissolve adequately in biological fluids, which can severely limit their absorption and therapeutic effectiveness. This issue is particularly prevalent among new chemical entities that are lipophilic in nature. Without sufficient solubility, even the most potent drug can fail to reach its target site in the body, rendering it ineffective.
Instability (Moisture, Oxidation, Light Sensitivity)
Chemical stability is another critical factor that can lead to the failure of a drug candidate. Many compounds are sensitive to environmental factors such as moisture, oxidation, and light. These instabilities can lead to degradation, resulting in reduced efficacy and safety concerns. For instance, a drug that degrades in the presence of moisture may lose its potency before it even reaches the patient, making stability a paramount consideration in F&D.
Low Bioavailability
Bioavailability refers to the proportion of a drug that enters the systemic circulation when introduced into the body. Low bioavailability can occur due to various factors, including poor solubility and extensive first-pass metabolism. Even if a drug is absorbed, its therapeutic effect may be diminished if it is rapidly metabolized or eliminated from the body. This challenge necessitates innovative F&D strategies to enhance bioavailability and ensure that patients receive the intended therapeutic benefits.
Inconsistent Release Profiles
Inconsistent release profiles can lead to unpredictable therapeutic outcomes. A drug that releases too quickly may cause toxicity, while one that releases too slowly may fail to achieve therapeutic levels. Achieving a consistent and controlled release profile is essential for maintaining drug efficacy and safety. This challenge underscores the importance of F&D in designing formulations that provide reliable and reproducible drug delivery.
F&D Resolves These Challenges Through:
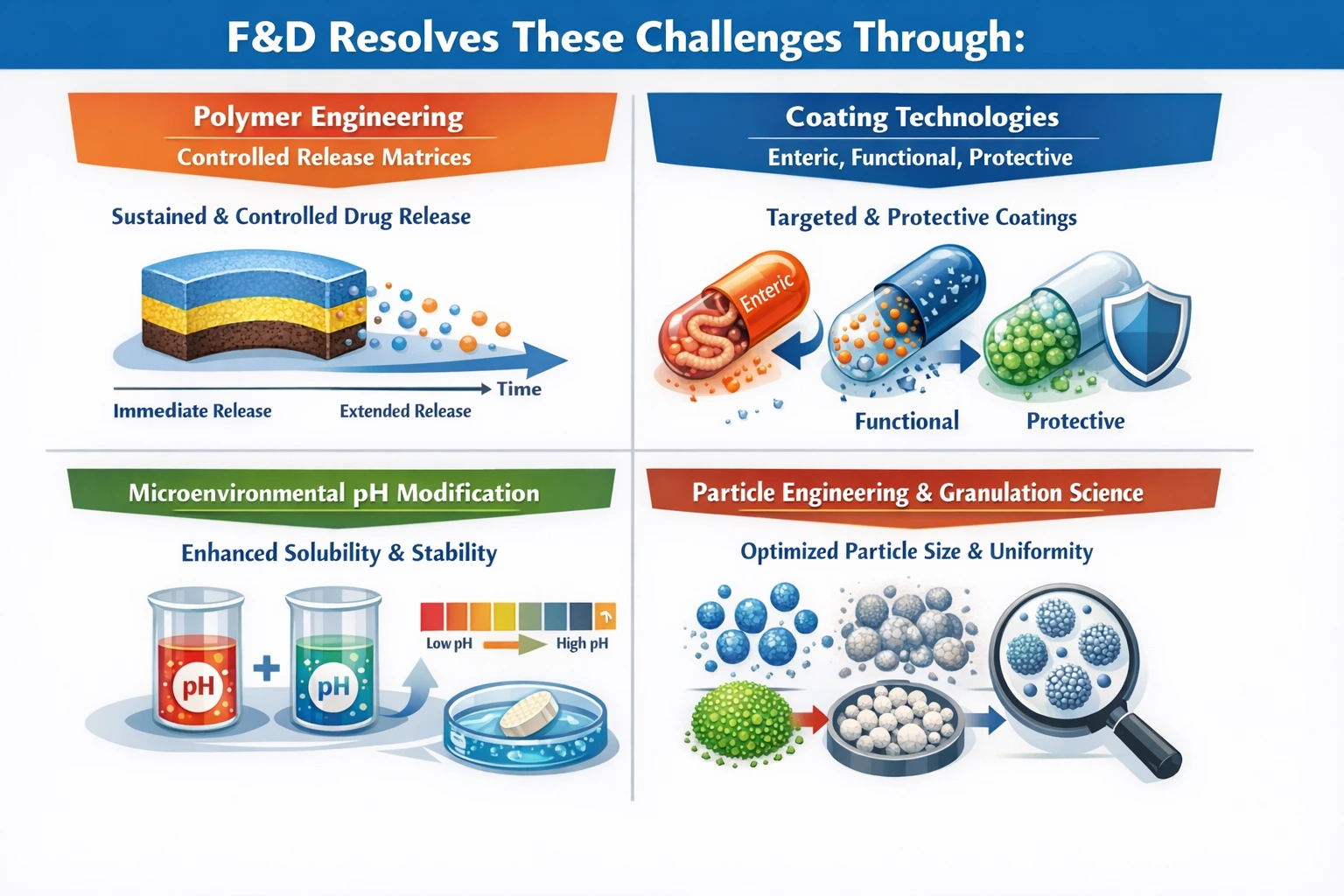
Polymer Engineering (e.g., Controlled Release Matrices)
One of the most effective strategies employed in F&D is polymer engineering. By utilizing controlled release matrices, formulators can design drug delivery systems that release active ingredients at a predetermined rate. This approach not only enhances bioavailability but also helps to mitigate the risks associated with inconsistent release profiles. Controlled release formulations can improve patient compliance by reducing the frequency of dosing, ultimately leading to better therapeutic outcomes.
Coating Technologies (Enteric, Functional, Protective)
Coating technologies play a vital role in protecting sensitive compounds from environmental factors and controlling their release profiles. Enteric coatings can prevent drug release in the acidic environment of the stomach, ensuring that the drug is released in the more neutral pH of the intestines. Functional coatings can enhance solubility and bioavailability, while protective coatings can shield drugs from moisture and oxidation. These technologies are essential for ensuring the stability and efficacy of pharmaceutical products.
Microenvironmental pH Modification
Modifying the microenvironmental pH can significantly enhance drug solubility and stability. By adjusting the pH of the formulation, formulators can create conditions that favor the dissolution of poorly soluble compounds. This strategy is particularly useful for drugs that exhibit pH-dependent solubility, allowing for improved absorption and bioavailability.
Particle Engineering and Granulation Science
Particle engineering and granulation science are crucial in optimizing the physical properties of drug formulations. By manipulating particle size and morphology, formulators can enhance solubility, improve flowability, and achieve desired release profiles. Granulation techniques can also improve the uniformity of the formulation, ensuring consistent dosing and therapeutic effects.
In Reality:
F&D is where theoretical success meets practical reality. While a molecule may exhibit excellent pharmacological properties in vitro, it is the F&D process that determines whether it can be successfully developed into a viable therapeutic product. The challenges of poor solubility, instability, low bioavailability, and inconsistent release profiles are not merely technical hurdles; they are critical factors that can dictate the success or failure of a drug candidate.

F&D is not just a support function; it is a strategic component of drug development that bridges the gap between laboratory research and clinical application. By addressing the multifaceted challenges that can impede drug efficacy, F&D plays an indispensable role in bringing innovative therapies to market. As the pharmaceutical landscape continues to evolve, the importance of F&D will only grow, underscoring its position as a cornerstone of successful drug development.
Conclusion
In the vast architecture of pharmaceuticals, discovery is only the first spark, delivery is what turns it into impact. The journey from R&D to F&D is not a linear handoff but a delicate, almost invisible choreography where molecules are not just studied, but taught how to survive, adapt, and perform within the human body.
A drug, no matter how brilliant in design, remains an unrealized promise until formulation gives it purpose. Without F&D, even the most elegant molecular innovation is reduced to potential; fragile, unstable, and ultimately unreachable. It is formulation that shields it from degradation, guides it through physiological barriers, and releases it with precision where and when it matters most.
But this is only the surface!
As medicine moves toward personalization, complexity deepens. Therapies are no longer built for populations, they are engineered for individuals. In this evolving landscape, F&D does not just support R&D; it begins to reshape it, influencing how molecules are designed from the very beginning. The boundary between discovery and delivery is dissolving, giving rise to a new paradigm where both are co-authors of therapeutic success.
And yet, the most intriguing part of this story remains ahead!
Because the future of pharmaceuticals is not just about better drugs or smarter formulations, it is about intelligent systems that respond, adapt, and communicate within the body itself. A future where delivery is no longer passive, but dynamic, almost alive!
References
- Allen, L. V., Popovich, N. G., & Ansel, H. C. (2020). Ansel’s Pharmaceutical Dosage Forms and Drug Delivery Systems (11th ed.). Wolters Kluwer. → Foundational reference for dosage form design and the role of formulation in drug delivery.
- Aulton, M. E., & Taylor, K. M. G. (2018). Aulton’s Pharmaceutics: The Design and Manufacture of Medicines (5th ed.). Elsevier. → Core text explaining the transition from drug substance to drug product.
- U.S. Food and Drug Administration (2006). Guidance for Industry: Q8(R2) Pharmaceutical Development. → Introduces Quality by Design (QbD) and emphasizes formulation as a critical component of product development.
- International Council for Harmonisation (2009). ICH Q8(R2): Pharmaceutical Development. → Regulatory framework linking R&D with formulation development and product performance.
- International Council for Harmonisation (2009). ICH Q9: Quality Risk Management. → Supports risk-based approaches in formulation and process development.
- Dressman, J., & Reppas, C. (2000). In vitro–in vivo correlations for lipophilic, poorly water-soluble drugs. European Journal of Pharmaceutical Sciences, 11(S2), S73–S80. → Highlights bioavailability challenges and the importance of formulation strategies.
- Amidon, G. L., Lennernäs, H., Shah, V. P., & Crison, J. R. (1995). A theoretical basis for a biopharmaceutic drug classification: The correlation of in vitro drug product dissolution and in vivo bioavailability. Pharmaceutical Research, 12(3), 413–420. → Introduces the BCS system—critical for understanding solubility and permeability limitations.
- Sinko, P. J. (2011). Martin’s Physical Pharmacy and Pharmaceutical Sciences (6th ed.). Lippincott Williams & Wilkins. → Covers physicochemical challenges like solubility, stability, and drug-excipient interactions.
- Hancock, B. C., & Parks, M. (2000). What is the true solubility advantage for amorphous pharmaceuticals? Pharmaceutical Research, 17(4), 397–404. → Discusses formulation strategies for poorly soluble drugs.
- Leuner, C., & Dressman, J. (2000). Improving drug solubility for oral delivery using solid dispersions. European Journal of Pharmaceutics and Biopharmaceutics, 50(1), 47–60. → Key reference for formulation-based bioavailability enhancement.
- Remington: The Science and Practice of Pharmacy (2021). Pharmaceutical Press. → Comprehensive reference for formulation science and drug delivery systems.
- Qiu, Y., Chen, Y., Zhang, G. G. Z., Yu, L., & Mantri, R. V. (2016). Developing Solid Oral Dosage Forms: Pharmaceutical Theory and Practice (2nd ed.). Academic Press. → Practical insights into formulation design, scale-up, and manufacturability.

