Somewhere in a hospital ward, a child lies in a bed with a fever. A nurse enters holding a small white tablet a single dose of medicine that, if it is exactly what it claims to be, will begin saving her life.
But how does anyone know?
How can anyone be certain that this tablet manufactured thousands of miles away, pressed by a machine producing hundreds of thousands of units per hour contains precisely the right molecule, in precisely the right amount, free from every harmful contaminant?
The answer is one word: analysis.
Pharmaceutical analysis is not a bureaucratic formality. It is not a box to be checked or overhead to be minimised. It is the bridge between the chemistry of a molecule and the healing it is meant to provide.
This essay traces the complete analytical journey of a pharmaceutical product: from raw material testing and in-process controls, through finished product release, across every dosage form
2: What Is Pharmaceutical Analysis — and Why Does It Matter?
Pharmaceutical analysis is the application of chemical, physical, biological, and microbiological methods to characterize, identify, quantify, and ensure the quality of pharmaceutical substances and products. It encompasses a vast range of techniques from the simplest visual inspection to the most sophisticated mass spectrometric analysis all unified by a single purpose: to confirm that a pharmaceutical product is what it claims to be, in the quantity it claims to contain, and free from any harmful impurities or contaminants.
The field draws from analytical chemistry, biochemistry, microbiology, statistics, and regulatory science. A pharmaceutical analyst must not only master the technical aspects of their instruments and methods but also understand the regulatory frameworks within which their work exists — primarily the pharmacopoeias (USP, BP, EP, JP, IP) and the guidelines of regulatory agencies such as the FDA (United States), EMA (Europe), and WHO.
The conceptual foundation of pharmaceutical analysis rests on four quality pillars, each associated with a specific class of analytical tests and a distinct regulatory obligation:
Table 2.1The Four Pillars of Pharmaceutical Quality and Their Primary Analytical Tests
| Pillar | Definition | Primary Analytical Tests |
|---|---|---|
| Identity | Confirmation that the substance is what its label states; detection of substitution, adulteration, or mislabelling | IR/FTIR, NIR, Raman, HPLC retention time, NMR, melting point, optical rotation |
| Purity | Confirmation that the substance is free from objectionable levels of impurities — related substances, residual solvents, elemental impurities, degradation products | HPLC gradient (related substances), GC-FID headspace (residuals solvents), ICP-MS (elemental impurities) |
| Potency (Assay) | Quantitative determination of actual API content relative to label claim; verification that potency falls within defined acceptance limits | HPLC (reversed-phase, ion-exchange), UV-Vis spectrophotometry, potentiometric titration, biological assay (for biologics) |
| Safety | Freedom from microbiological contamination, endotoxins, particulate matter, toxic elemental impurities, and any substance capable of causing harm at likely exposure | TAMC/TYMC , Sterility (USP <71>), LAL/BET , ICP-MS , Particulate matter |
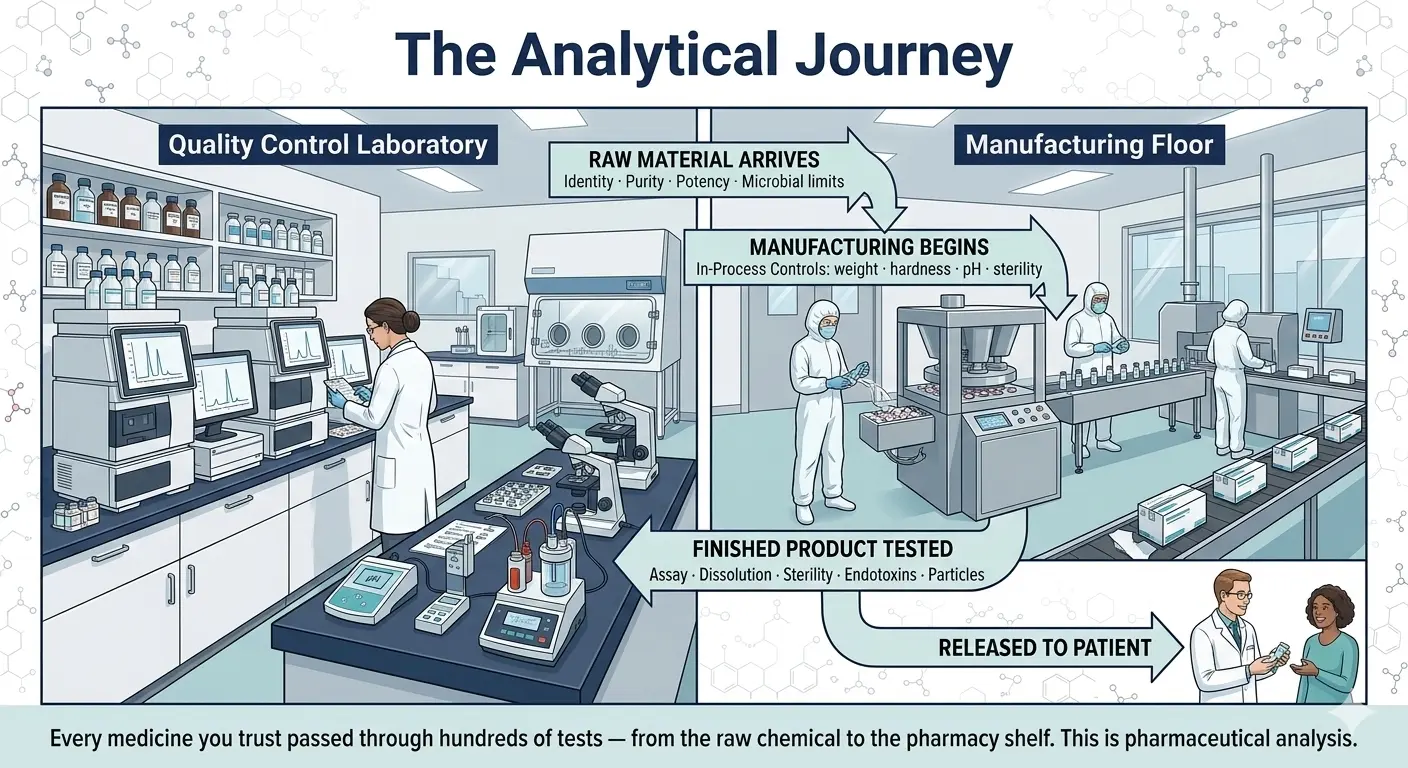
3: The Cycle of Analysis — From Raw Material to Finished Product
The analytical cycle in pharmaceutical manufacturing follows the product through three distinct stages: Raw Material Analysis, In-Process Control (IPC), and Finished Product Analysis. Each stage has specific objectives, tests, and acceptance criteria. Together, they form a continuous quality assurance cycle that accompanies the product from its arrival at the manufacturing site to its release for patient use.
3.1 Raw Material Analysis — The Foundation of Quality
Every pharmaceutical product begins with raw materials. These include the Active Pharmaceutical Ingredient (API) — the molecule responsible for the therapeutic effect — and excipients, the inactive ingredients that give the dosage form its physical form and properties. Raw materials arrive at the manufacturing site from external suppliers and must be thoroughly analyzed before they can be released for use in production.
The principle is simple but critical: garbage in, garbage out. No manufacturing process, however well controlled, can produce a quality product from substandard starting materials. Raw material analysis is therefore the first and arguably most important gate in pharmaceutical quality control.
3.1.1 Raw Material Testing — Key Analyses
A. Identification Testing
Purpose: To confirm that the received material is what it is claimed to be. This is performed on every individual container received.
Infrared Spectroscopy (FTIR/ATR-FTIR): The gold standard for API identification. Each molecule produces a unique IR spectrum a ‘molecular fingerprint’ by absorbing infrared radiation at specific wavelengths corresponding to its chemical bonds. The spectrum of the sample is compared to a reference standard spectrum. For ATR-FTIR (Attenuated Total Reflectance), the sample requires no preparation it is simply pressed against a crystal. Identification passes if the sample spectrum matches the reference within specified correlation limits (typically R² > 0.999).
Raman Spectroscopy: Complementary to FTIR, providing information about molecular vibrations that are IR-inactive. Useful for materials that absorb water (where FTIR can be problematic) and for identification through packaging (handheld Raman devices can identify materials without opening containers). Principle: inelastic scattering of monochromatic laser light by molecular bonds.
HPLC Retention Time: In quantitative assay methods, the retention time of the sample peak compared to a reference standard serves as a confirmation of identity. Not used as the sole identity test but provides additional confirmation.
B. Assay (Potency) Testing
Purpose: To determine the actual content of the API or active excipient in the received material, ensuring it meets the specified potency range (e.g., 98.0–102.0% ).
High-Performance Liquid Chromatography (HPLC): The most widely used technique for pharmaceutical assay. The sample is dissolved in a suitable solvent and injected into a column packed with a stationary phase (typically C18 reversed-phase silica). A mobile phase (usually a mixture of water, organic solvents, and buffers) carries the analyte through the column. Components are separated based on their differential interactions with stationary and mobile phases. Detection is most commonly by UV absorption (UV-DAD detector) or by mass spectrometry (LC-MS). The peak area of the analyte is compared to that of a reference standard to calculate the percentage content. HPLC is specific, accurate, and capable of simultaneous assay and impurity profiling.
UV-Visible Spectrophotometry: Used for simpler assays where specificity is less critical or for compendial methods. Based on Beer-Lambert Law: A = ε × c × l, where A is absorbance, ε is the molar absorptivity coefficient, c is concentration, and l is path length. The sample is dissolved in a solvent, and absorbance is measured at the λmax (wavelength of maximum absorption) of the analyte against a reference standard or using a standard curve. Less specific than HPLC since it cannot distinguish between the API and co-eluting impurities. Fast and inexpensive.
Titration Methods: Classical wet chemistry methods still used for certain APIs and excipients. Acid-base titration (for acids, bases, and salts), complexometric titration with EDTA (for metal-containing compounds), and redox titration are all employed. Modern pharmaceutical laboratories use automated potentiometric titrators that use pH electrodes or other potentiometric sensors to detect the endpoint precisely, eliminating the subjectivity of visual indicators.
C. Water Content (Moisture) Determination
Karl Fischer Titration (KFT): The definitive method for water determination in pharmaceutical materials. Based on the stoichiometric reaction of water with iodine, sulfur dioxide, and a base in the presence of an alcohol (the Karl Fischer reagent). In volumetric KF, the sample is titrated with a standardized KF reagent to an electrometric endpoint. In coulometric KF (for very low water contents, <1%), iodine is generated electrochemically. Water content is critical because excess moisture promotes hydrolytic degradation of APIs and microbial growth in excipients.
Loss on Drying (LOD): A simpler but less specific method. The sample is weighed, dried at a specified temperature (typically 105°C) for a defined time or until constant weight, then re-weighed. The % LOD = (weight loss / initial weight) × 100. Measures not only water but also any other volatile substances. Used where KFT is not applicable and as a quick in-process test.
D. Impurity Profiling
Purpose: To detect and quantify related substances (process impurities, degradation products, and isomers) that may be present in the API.
HPLC with UV-DAD or MS Detection: The primary tool for related substances testing. A gradient HPLC method is typically used to elute both the API and its impurities from the column over a programmed mobile phase gradient. Individual impurities are quantified against either the API peak (using relative response factors) or against individual impurity reference standards. ICH Q3A specifies reporting, identification, and qualification thresholds. Mass spectrometric (LC-MS/MS) detection provides structural information for unknown impurity identification.
Gas Chromatography (GC): The method of choice for residual solvent testing (ICH Q3C). Solvents used in API synthesis (e.g., acetone, methanol, toluene, dichloromethane) must be tested and shown to be below their ICH class limits. In headspace GC, the sample is dissolved in DMSO or water, and the vial headspace above the solution is injected into the GC. Separation occurs on a capillary column (typically DB-624 or DB-5), and detection is by FID (Flame Ionization Detector). Static headspace avoids introducing non-volatile materials into the GC column.
E. Elemental Impurity Testing
ICP-MS (Inductively Coupled Plasma — Mass Spectrometry): The most sensitive and comprehensive technique for elemental impurity analysis, required by ICH Q3D. The sample is digested in nitric acid (microwave digestion) and the resulting solution is introduced into a plasma torch (Ar plasma at ~10,000 K). Elements are ionized and detected by mass spectrometry at their characteristic mass-to-charge ratios. ICP-MS can detect elements at parts-per-trillion (ppt) levels simultaneously for 30+ elements, including lead, cadmium, arsenic, mercury (Class 1 elements), and platinum, palladium, iridium (Class 2A catalyst residues).
ICP-OES (Inductively Coupled Plasma — Optical Emission Spectrometry): Similar plasma ionization but detection by optical emission rather than mass spectrometry. Less sensitive than ICP-MS (ppb rather than ppt) but faster and more robust for higher concentration ranges. Used for Class 2B and Class 3 elemental impurities.
F. Microbiological Quality of Raw Materials
Total Aerobic Microbial Count (TAMC) and Total Yeast/Mold Count (TYMC): Performed on non-sterile API and excipients. The sample is dissolved or suspended in buffer and filtered through a 0.45 µm membrane filter or plated directly. TAMC uses Tryptic Soy Agar (TSA) incubated at 30-35°C for 3-5 days. TYMC uses Sabouraud Dextrose Agar (SDA) incubated at 20-25°C for 5-7 days. USP/EP limits vary by material type (e.g., APIs for non-aqueous dosage forms: TAMC ≤10³ CFU/g, TYMC ≤10² CFU/g).
Specified Organism Testing: Detection of specific harmful organisms (E. coli, Salmonella spp., Staphylococcus aureus, Pseudomonas aeruginosa, Clostridium spp.) in certain raw materials as required by the pharmacopoeia based on the route of administration of the final product.
G. Physical and Other Tests
| Test | Method/Instrument | Purpose |
| Particle Size Analysis | Laser Diffraction (Malvern Mastersizer) / Sieve Analysis | Controls dissolution rate and content uniformity of the API |
| Bulk & Tapped Density | Tap Density Tester (Jolting Volumeter) | Critical for powder flow, die fill uniformity in tablet press |
| Specific Surface Area | BET (N₂ gas adsorption) | Affects dissolution; critical for poorly soluble APIs |
| Melting Point / DSC | Differential Scanning Calorimetry (DSC) | Confirms identity and polymorphic form; detects impurities |
| Polymorphism | XRPD (X-Ray Powder Diffraction) | Different crystal forms can have vastly different solubility |
| pH (in solution) | Calibrated pH meter (glass electrode) | Affects stability, solubility, and membrane permeation |
| Heavy Metals (limit test) | ICP-MS / Colorimetric (Ph.Eur. 2.4.27) | Detects toxic metallic impurities within safety limits |
| Color & Appearance | Visual / Colorimetric comparison | First quality indicator; flags contamination or degradation |
| Optical Rotation | Polarimeter | Confirms chirality of optically active APIs (e.g., amino acids) |
| Refractive Index | Refractometer | Identity/purity test for liquid excipients and APIs |
3.2 In-Process Controls (IPC): Activities, Parameters, and Manufacturing Stage Examples
What Are IPCs?
In-process controls are analytical tests and physical measurements performed at defined intervals during manufacturing to confirm that the process remains within validated parameters. they are specific to each manufacturing step and dosage form. Unlike release testing, IPCs enable real-time intervention, preventing batch failure before it propagates across thousands of units.
Common IPC Tests and Parameters
| IPC Test | What Is Monitored | Why It Matters |
|---|---|---|
| Weight Variation | Individual unit weight vs. target | Reflects die fill consistency; indirectly predicts content uniformity |
| Hardness / Crushing Strength | Force required to fracture tablet (Newtons) | Controls disintegration time, dissolution rate, and resistance to handling |
| Thickness | Tablet or capsule dimensions (mm) | Critical for blister packaging fit; indirect measure of compression force |
| Friability | % weight loss after mechanical stress | Predicts tablet survival during packaging, transport, and dispensing |
| Disintegration Time | Time for complete tablet/capsule breakup at 37°C | Prerequisite for dissolution and drug absorption |
| pH Measurement | Hydrogen ion concentration of solution/suspension | Controls chemical stability (hydrolysis rates are pH-dependent) and patient acceptability |
| Viscosity | Resistance to flow (mPa·s or cP) | Governs pourability, fill volume accuracy, and suspension homogeneity |
| Moisture Content (LOD/KF) | % water or volatile content in powder/granule | Prevents hydrolytic degradation, microbial growth, and capping/lamination defects |
| Blend Uniformity | API distribution across blender volume (% RSD) | Ensures consistent dose in every unit; %RSD must be ≤5.0% across ≥10 sample locations |
| Particle Size Distribution | d10, d50, d90 of granules or API | Controls flow, compressibility, dissolution rate, and content uniformity |
| Content Uniformity | Individual unit API content by HPLC | Confirms homogeneous API distribution — more sensitive than weight variation alone |
| Clarity / Appearance | Visual or turbidimetric inspection | Detects precipitation, particulates, or phase separation in liquids |
| Density (Specific Gravity) | g/cm³ of liquid preparation | Confirms correct concentration and fill volume accuracy |
| Filter Integrity | Bubble point pressure (bar) or forward flow (mL/min) | Confirms the sterilising filter is intact before and after sterile filtration |
| Fill Volume / Fill Weight | Volume or mass per filled container | Ensures each patient receives the correct dose |
| Bioburden (Pre-sterilisation) | CFU/mL of bulk solution before sterilisation | Validates the pre-sterilisation microbial load is within the validated process range |
4.Dosage Form-Specific Analysis ( Summary)
4.1 Tablets
| Test | How Performed |
|---|---|
| Hardness | Diametral crushing force (hardness tester, N) |
| Friability | Rotating drum friabilator; 100 rotations; % weight loss |
| Disintegration | Disintegration tester; specified medium; 37°C; time to complete breakup |
| Dissolution (IR) | USP App. 2 paddle; 900 mL; 37°C; samples by HPLC/UV |
| Dissolution (ER) | USP App. 1 or 3; multi-point sampling; full profile |
| Dissolution (Enteric) | Stage 1: pH 1.2 HCl (2 h, must stay intact) → Stage 2: pH 6.8 buffer; HPLC/UV |
| Content Uniformity | 10 tablets individually dissolved and assayed by HPLC |
| Weight Variation | 20 tablets individually weighed; deviation from mean |
| Moisture Content | Karl Fischer titration or Loss on Drying (105°C) |
| Water Activity | Water activity meter |
| Microbial Limits | TSA (TAMC) and SDA (TYMC) plate count; specified organism enrichment culture |
4.2 Capsules
| Test | How Performed |
|---|---|
| Disintegration | Disintegration tester; enzyme added if HPMC or cross-linked gelatin |
| Dissolution (standard) | USP App. 1 basket with sinker; HPLC/UV |
| Dissolution (2-stage) | Stage 1 without enzyme → if failure, repeat with pepsin or pancreatin added |
| Assay / Content Uniformity | Contents emptied; dissolved; protein precipitation if needed; HPLC |
| Net Fill Weight | Gross weight minus empty shell weight |
| Water Content (fill) | Karl Fischer coulometric titration |
| Oxygen Headspace (softgels) | Headspace GC or laser headspace analyser on sealed capsule |
| Shell Thickness (softgels) | Micrometer at defined positions |
| Leaker Test (softgels) | Vacuum immersion in methylene blue; inspect for dye ingress |
4.3 Injectables
Sterility
| Method | How Performed |
|---|---|
| Membrane Filtration | 0.45 µm filtration; membrane to FTM (30–35°C) + SCDM (20–25°C); 14 days; turbidity = fail |
| Direct Inoculation | Product inoculated directly into both media; 14 days; used for oily/viscous products |
Endotoxin Testing
| Method | How Performed |
|---|---|
| Gel-Clot | Sample + LAL reagent; 37°C 60 min; gel formation = positive |
| Turbidimetric | Turbidity monitored kinetically; time to threshold vs. standard curve |
| Chromogenic | Absorbance at 405 nm; p-nitroaniline released proportional to endotoxin |
| rFC Assay | Recombinant Factor C; fluorescent substrate; no horseshoe crab lysate |
| MAT | Human monocyte challenge; cytokine release by ELISA; detects all pyrogens |
| Rabbit Pyrogen Test | IV injection to 3 rabbits; rectal temperature monitored 3 h |
Particulate Matter
| Test | How Performed |
|---|---|
| Visible | 100% visual inspection under 2,000–3,750 lux; black and white backgrounds |
| Sub-visible | Light obscuration (HIAC); ≥10 µm and ≥25 µm channels |
| Protein Aggregates | MFI (imaging), DLS (hydrodynamic size), SEC-HPLC (monomer vs. aggregate) |
Other Injectable Tests
| Test | How Performed |
|---|---|
| pH | Two-point calibrated pH meter |
| Osmolarity | Freezing-point depression osmometry |
| CCI | Vacuum decay; HVLD; or headspace laser spectroscopy |
| Extractables/Leachables | GC-MS and LC-MS on container extracts |
| Deliverable Volume | Contents emptied; measured in calibrated cylinder |
| Residual Moisture (lyophilisates) | Karl Fischer coulometric titration |
| Reconstitution Time (lyophilisates) | Timed to complete dissolution; visual check for particles |
4.4 Semi-Solids
| Test | How Performed |
|---|---|
| Assay | API extracted from base; HPLC (lipophilic base: solvent extraction → aqueous back-extraction) |
| pH | 10% w/w aqueous dispersion; calibrated pH meter |
| Viscosity / Rheology | Brookfield viscometer or oscillatory rheometer (G’, G”) |
| Particle Size | Laser diffraction (Malvern); d10, d50, d90 |
| Drug Release (IVRT) | Franz diffusion cell; synthetic membrane; receptor phase by HPLC; flux vs. √time |
| Phase Stability | Centrifugation 5,000 rpm / 30 min; accelerated storage 40°C/75% RH |
| Preservative Assay | HPLC against reference standard |
| Metal Particles | Melted preparation on glass slide; 30× microscopy; USP <751> |
4.5 Oral Liquids
| Test | How Performed |
|---|---|
| Assay | HPLC; suspensions fully resuspended before sampling |
| pH | Calibrated pH meter on undiluted product |
| Density | Oscillating U-tube densitometer (Anton Paar) |
| Viscosity | Brookfield viscometer; appropriate spindle and speed |
| Alcohol Content | Headspace GC or direct GC-FID |
| Preservative Assay | HPLC against reference standard |
| Particle Size (suspensions) | Laser diffraction; d90 reported |
| Resuspendability | Manual inversion; time to homogeneity recorded |
| Zeta Potential (emulsions) | Laser Doppler microelectrophoresis (Zetasizer) |
| Extractable Volume | Full contents emptied per label; measured in calibrated cylinder |
| Sedimentation Volume | F = V_sed / V₀; calculated after defined settling period |
4.6 Inhalation Products
| Test | How Performed | Product |
|---|---|---|
| Delivered Dose Uniformity | Dose collected at beginning, middle, and end of device life; HPLC assay | MDI, DPI |
| APSD / FPD | NGI or ACI cascade impaction; HPLC per stage | MDI, DPI |
| Spray Pattern | Impaction paper or laser sheet imaging; ovality ratio measured | MDI |
| Leakage Rate | Device weighed; stored inverted 30 days; reweighed | MDI |
| Propellant Content | Headspace GC; HFA 134a or HFA 227ea quantified | MDI |
| Moisture Content | Karl Fischer coulometric titration | DPI |
| Particle Size | Laser diffraction on nebulised aerosol; VMD and % <5 µm | Nebuliser |
4.7 Suppositories
| Test | How Performed |
|---|---|
| Weight Variation | Each unit individually weighed |
| Melting / Softening Time | 37°C water bath apparatus; time to complete deformation |
| Disintegration | pH 7.0 buffer at 37°C; disintegration apparatus |
| Drug Release | USP App. 2 paddle; HPLC on withdrawn samples |
| Assay | Dissolved in solvent (lipophilic: chloroform → aqueous back-extraction); HPLC |
| Hardness | Texture analyser; probe penetration force (N) |
4.8 Ophthalmics
| Test | How Performed |
|---|---|
| Sterility | Membrane filtration (solutions); direct inoculation (ointments); 14 days |
| Particulate Matter | Light obscuration; tighter limits than USP <788> |
| pH | Calibrated pH meter on undiluted product |
| Osmolarity | Freezing-point osmometry |
| Preservative Assay | HPLC against reference standard |
| Antimicrobial Effectiveness | Organism challenge; viable counts at days 2, 7, 14, 28 |
| Metal Particles | Melted preparation on glass slide; 30× microscopy |
4.9 Transdermal Patches
| Test | How Performed |
|---|---|
| Assay / Content Uniformity | Entire patch extracted in solvent; HPLC; 10 individual patches for CU |
| IVRT | USP App. 5 or 6; synthetic membrane; receptor phase by HPLC; flux vs. √time |
| IVPT | Franz cells; excised human skin; drug permeation into receptor phase by HPLC |
| Peel Adhesion | 180° peel on tensile tester; force in N/cm |
| Probe Tack | Texture analyser; probe contact and withdrawal force |
| Shear Resistance | Hanging weight parallel to adhesive surface; time to failure |
| Residual Drug Content | Used patch extracted; HPLC; confirms actual drug delivered |
| Cold Flow | Vertical storage; inspect for adhesive migration beyond patch border |
The Circle Closes Analysis as the Soul of Pharmaceutical Science
We began with a child in a hospital bed and a small white tablet. We traced that tablet backward through the pharmacy shelf, through distribution, through finished product testing, through manufacturing controls, through raw material analysis and at every stage, we found the same thing: analysis.
What this essay has covered represents only a structured overview. Behind every test described here lies years of method development, validation studies, reference standard certification, stability programmes, regulatory submissions, and analyst training. Behind every passing result is a system quality management, data integrity controls, inter-laboratory comparisons, regulatory inspections, and post-market surveillance ensuring that the number on the certificate of analysis reflects the truth in the patient’s hand.