Safety strategy forms the backbone of minimizing occupational exposure to hazardous drug products. Potential occupational risks and adverse effects on healthcare workers arise from exposure to hazards at the workplace. Exposure to hazardous drugs has adverse impacts on health, such as an increase in the risk of cancers, a risk of damage to organs, and/or reproductive risk.
Healthcare entities can make it possible to decrease such risks by the ongoing implementation of a risk management plan. An efficient and effective risk management plan requires the essential implementation of the following fundamentals: hazard identification, exposure assessment, risk assessment, and risk management.
A full risk management plan is concerned with the engineering controls, administrative controls, and personal protective equipment (PPE) that will be implemented to mitigate the risks identified in the risk assessment. Risk management should also include periodic exposure assessments along with medical surveillance conducted to determine the degree of control fulfilled. The designed risk management plan should be only a part of a comprehensive Safety Management System. Several approaches have been developed in healthcare entities to ensure the protection of healthcare workers from occupational exposure to hazardous drug products.
In general, these approaches adhere to the hierarchy of controls for standard industrial hygiene practice. Such controls comprise the hazard elimination or substitution (if possible), accompanied by the use of engineering controls, administrative controls, and PPE. Engineering controls apply to modifications or interventions in the workplace environment designed to reduce or exclude healthcare workers’ exposure to potential hazards. Properly designed engineering controls are well integrated into daily tasks to provide a high level of protection.
Administrative controls are procedures implemented by healthcare organizations to minimize the risk of exposure to workplace hazards. Further on, they focus on managing or altering daily tasks instead of changing physical conditions or depending on protective equipment. PPE provides worker protection to reduce exposure to hazardous drugs. Personal Protective Equipment serves as the last line of defense against protecting healthcare workers against potential hazards. It is important to understand the proper use and limitations of any selected PPE to ensure that it is suitably fitted and is constructed of materials that protect from exposure to hazardous drug products.
Medical surveillance is a part of the general exposure control program, harmonizing with engineering controls, safe work processes, administrative controls, and the use of PPE. All healthcare workers who are involved in handling hazardous drug products should be enrolled in the medical surveillance program. Such a program ensures the mitigation of the impact of adverse health effects on health workers who are potentially exposed to hazardous drug products.
Risk Assessment
Periodic follow-up assessments should be conducted when any deviations from those assigned control practices are noted. Generally, the potential risks to healthcare workers are evaluated by considering the following factors:
– The potential hazards associated with handling hazardous products.
– The likelihood of the occurrence of adverse health impacts.
– The severity of the health effects.
The potential hazards associated with handling the hazardous drugs mentioned in the occupational exposure assessment were previously described. The evaluation of the dosage form and identification of the probability of exposure should be included in the risk assessment.
Likelihood of Occurrence
Evaluation of the likelihood that a certain identified exposure would result in harm to workers is conducted. Consideration of the dosage form and packaging of the material in the workplace, attempted activities, frequency of exposure, and engineering controls in the workplace are considered in this evaluation. Exposure monitoring should help to determine the potential risk mitigation interferences.
Safety strategy: Severity of Health Effects
NIOSH urges the consideration of worst-case scenarios, through the identification of the maximum concentration that a healthcare worker may be exposed to and the potential impact it has on health. Identifying the areas, processes, and scenarios of concern, and the creation and implementation of a facility-specific risk management plan for the safe handling of hazardous drug products should be included in the risk assessment. The risk management plan should establish workplace policies and procedures specific to the handling of hazardous drug products according to the hierarchy of controls.
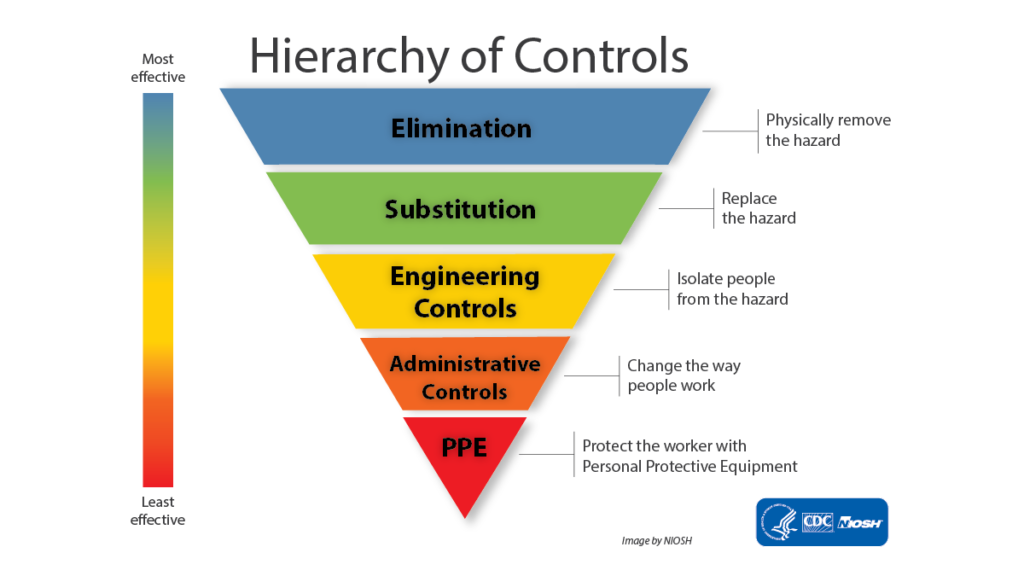
The hierarchy of controls (Figure 1)
The risk management plan or the hazard communication program enlightens the hazard identification, exposure assessment, and risk assessment. Moreover, it identifies the features of intervention to mitigate the impact of exposures and reduce risks to healthcare workers. The plan typically comprises techniques of implementing engineering controls, administrative controls, PPE, periodic exposure assessments, and medical surveillance based on the sources of exposure and the risks identified in the risk assessment.
However, healthcare workers may be exposed to various hazardous drugs, sometimes on a daily basis over years, and the combined effects of hazardous drugs are hardly studied. In general, the recommendations follow the hierarchy of controls (Figure 1) for standard hygiene procedures that include elimination or substitution, whenever possible, and the use of engineering controls, administrative controls, and PPE.
The controls listed at the head of the hierarchy are the most effective and provide the best business value. Such controls are designed to offer lower healthcare workers’ compensation rates to lower healthcare costs to care for harmed workers, to improve operational efficiency, and decrease employee absences and turnover. A written plan that details how the healthcare institutions will implement the hierarchy of controls helps all healthcare workers, supervisors, and supporting departments understand their relevant responsibilities and roles for handling hazardous drugs safely
Those institutions are required to develop policies and procedures that ensure healthcare worker safety during all situations of hazardous drug product handling. The institution must develop SOPs to ensure effective training regarding proper labeling, transport, storage, and disposal of hazardous drugs and use of SDS, based on the Globally Harmonized System of Classification and Labeling of Chemicals.
The hazard communication program plan
A written plan that describes how the procedure will be implemented; as it should be written and addressed to all healthcare workers. The plan should deal with all aspects of how to safely handle hazardous drugs throughout the facility in terms of receiving, compounding, storage, handling of waste, and spill control. The written risk management plan should be part of an overall Safety Management System implemented to manage all safety risks in the workplace.
- All containers of hazardous drugs must be labeled.
- Entities must have an SDS for each hazardous drug used (29 CFR 1910.1200).
- Entities must ensure that the SDSs for each hazardous drug used are easily accessible to personnel during working hours.
- Personnel who may be exposed to hazardous drugs when working must be given detailed information and training before the first task to work with a hazardous drug, and whenever there is a hazard change.
- Personnel of reproductive capability must provide a signed approval that they understand the risks of handling hazardous drugs.
Engineering Controls
Engineering controls are implemented to reduce exposures by separating a hazardous drug from the workplace environment, such as by ventilation or by locating a barrier between the worker and the hazard. Barriers typically isolate the process or equipment to contain the hazard and hence prevent the hazard from entering the workplace environment. Well-designed engineering controls are routinely independent of daily interactions to provide a high level of protection. Engineering controls include, but are not limited to, biological safety cabinets (BSCs) and compounding aseptic containment isolators. Engineering controls require proper installation, inspection, preventive maintenance, and repair. These controls should be regularly disinfected, deactivated, cleaned, and cleaned.
Formulations of hazardous drugs, namely tablets, capsules, powders, transdermal dosage forms, and liquid, sterile or nonsterile, should be dealt with inside a BSC or CACI designed to prevent the release of the hazard into the work environment. Certain areas are well taken care of to be maintained at negative pressure relative to the surrounding areas to contain hazardous drugs and mitigate the potential risk due to exposure. Consideration should be given to providing uninterrupted power sources for the ventilation systems to sustain the negative pressure in case of power loss.
Use a HEPA filter, all over the entire workplace, for the exhaust, and preferably exhaust 100% of the filtered air to the outside. Ensure that the air exhausted outside is not pulled back into the building by the heating, ventilation, and air conditioning (HVAC) systems or by the windows, doors, or other points of entry. Use fans downstream of the HEPA filter to ensure that the contaminated ducts are maintained under negative pressure.
Administrative Controls
Administrative controls are most effective when they are supported by a safety and health culture within an organization. They function through the reduction of the airborne and surface concentration of workplace contaminants and so can reduce workers’ potential exposure. Healthcare Workers should receive training and prove competency before the initiative works with hazardous drugs. All personnel who handle hazardous drugs must be well trained based on their job functions (e.g., in the receipt, storage, compounding, repackaging, dispensing, administering, and disposing of HDs).
Personnel competency must be annually reevaluated. Personnel must be trained before the introduction of new hazardous drugs or new equipment and before any change in the procedures. Documentation of all training and competency assessments must be performed. The training must minimally include the following:
- Overview of the entity’s list of HDs and their risks.
- Review of the entity’s SOPs related to the handling of HDs.
- Proper use of PPE
- Proper use of equipment and devices (e.g., engineering controls).
- Response to known or suspected HD exposure.
- Spill management.
- Proper disposal of HDs and trace-contaminated materials.
Other administrative controls should ensure the following approaches:
- Limit the operation time during which workers deal with hazardous drugs. Limit access to the areas where hazardous drugs are handled. Implement good cleaning practices (such as using dedicated cleaning tools for areas where workers use hazardous drugs, wet wipes for cleanup, and HEPA-filtered vacuums). Mandate handwashing with soap and water (preferred to apply alcohol after washing) before eating, drinking, smoking, using the bathroom, removing or applying cosmetics, or leaving the workplace. Prohibit the consumption of any food and continuously monitor compliance with policies and procedures, and work out any causal factors for non-compliance.
- Prohibit the use of unventilated areas, such as storage closets, for drug storage or any tasks involving hazardous drugs. Work surfaces should be deactivated, decontaminated, and cleaned before and after each activityDisposal of cleaning materials should follow the facility\’s SOPs.
- Throwing away of hazardous drugs is be done carefully to avoid spillage, and certain specified equipment are to be utilized with these drugs.
Personal Protective Equipment
PPE provides a barrier of protection to healthcare personnel to reduce the impact of exposure to hazardous drugs, aerosols, and residues. Healthcare workers should use PPE according to the context of a documented PPE program, which entitles them to initial training, retraining, and periodic testing of workers’ knowledge of the proper use of PPE. Disposable PPE must not be reused. Reusable PPE must be decontaminated and cleaned after use. Gowns, head, hair, shoe covers, and two pairs of chemotherapy gloves are required for handling sterile and nonsterile hazardous drugs.
For all other activities, the entity’s SOP must describe the appropriate PPE to be worn based on its occupational safety plan and assessment of risk. The entity must develop SOPs for PPE based on the risk of exposure and activities performed. OSHA requires the selection of PPE based on an assessment of workplace hazards. Proper understanding of the attributes and limitations of any selected PPE should be ensured to provide good protection, along with proper usage by workers handling hazardous drugs. Before using, workers should examine all PPE for any defects or damage. See figure 2

Deactivating, Decontaminating, Cleaning, and Disinfecting
All workplace areas where hazardous drugs are handled and all non-disposable equipment and devices must be deactivated, decontaminated, and cleaned. Moreover, sterile compounding areas and devices must be then disinfected. The organization should develop written procedures for decontamination, deactivation, and cleaning, and for disinfection. All personnel who perform deactivation, decontamination, cleaning, and disinfection activities in areas of hazardous drug handling must be well-trained according to relevant procedures to protect themselves and the environment from contamination.
All personnel performing these activities must wear appropriate PPE that is known to be resistant to the cleaning agents used at all stages, including two pairs of chemotherapy gloves and impermeable disposable gowns. In addition to eye protection and face shields. If warranted by the activity, respiratory protection must be used. Selection of the deactivating, decontaminating, cleaning, and disinfecting agents should be relevant and compatible with the type of hazardous drug contaminant(s), location, and surface materials.
Direct application of agents used for the deactivation, decontamination, and cleaning must be done through the use of wipes wetted with proper solution and never to be delivered by a spray bottle in order not to cause any spreading of the hazardous drugs residue. All disposable materials must be thrown away to meet EPA regulations and the organization’s policies. Perform cleaning in areas that are sufficiently ventilated.
| Cleaning stage | objective | Agents used |
| Deactivation | Make a hazardous compound inert or inactive | EPA-registered oxidizers e.g. peroxide or sodium hypochlorite |
| Decontamination | To get rid or clear hazardous drug residue | Alcohol, water, peroxide or sodium hypochlorite |
| Cleaning | To get rid or clear any organic or inorganic material | Germicidal detergent |
| Disinfection | To demolish any expected microorganism ( for areas designed to be sterile) | EPA- registered disinfectant and/or alcohol |
Spill Control
Only experienced well trained personnel are permitted to clean up a spill of hazardous drugs. Spills must be properly contained and cleaned immediately only by qualified trained personnel with appropriate PPE. Qualified personnel must be reachable at all times wherever hazardous drugs are being handled. Signs must be available for restricting access to the spill area. All spill materials must be classified and disposed of as hazardous waste. SOPs must address the size and scope of the spill and specify the responsible personnel for spill management and the type of PPE required.
Hazardous Drug Waste
As a rule, healthcare facilities have various controlled waste streams, including medical waste and hazardous drug waste, which can be categorized into three groups:
1. Resource Conservation and Recovery Act (RCRA) hazardous waste; where the drug waste meets the definition of a U.S. EPA RCRA hazardous waste (P-listed, U-listed, or D-coded), which is typically contained in a black container and managed by incineration at a permitted hazardous waste facility. Any RCRA hazardous waste containers should be kept closed at all times when not in active use, labeled appropriately according to both state and federal regulations, and moved to a central accumulation area when full.
2. Trace chemotherapy waste: Ampules, empty vials, syringes, trace-contaminated PPE, swabs, pads, etc., contained in a yellow hard-plastic container or a yellow hamper for soft items labeled as chemotherapy and biohazardous waste and managed by incineration at a regulated medical waste incinerator. Trace chemotherapy waste should be closed and sealed according to established practice for regulated medical waste and stored in the regulated medical waste area of the facility
3. Nonhazardous pharmaceutical waste: Drug waste that surpasses the criteria explained above is contained in a white or blue container and managed by incineration at any incinerator permitted for nonhazardous pharmaceutical waste
Follow-Up Plan
Any occurrence of exposure-related health changes should provoke immediate re-evaluation of the primary specified preventive measures (e.g., administrative and engineering controls, PPE, and others). As such, medical surveillance serves as a check on how effective the controls already in use are. The following actions should be accurately followed:
- Perform a post-exposure examination adapted to the type of exposure (e.g., spills or needle sticks from syringes containing hazardous drugs) where an assessment of the extent of exposure is conducted and included in a confidential database and an incident report along with physical examination Treatment and laboratory studies will follow the guidance as named by emergency protocols
- Compare the performance of controls with approved standards.
- Check and document that the built engineering controls are in proper operating condition
- Verify and document that the worker adheres to the stated policies. Regularly review policies for the use of PPE and personnel compliance with PPE use and policies. Always check the availability of appropriate PPE
- Develop and document a plan of action that will prevent any further potential exposure of workers
- Provide and document a follow-up medical survey that clearly evidence that the implemented plan is effective
- Guarantee that any exposed worker receives confidential notification of any adverse health effect and, if possible, offer an alternative task or temporary reassignment
- Provide an ongoing medical surveillance of all workers at potential risk of exposure to hazardous drugs to assess the effectiveness of the plan implemented.
References
Protecting Workers from the Hidden Dangers of Hazardous Drugs in Pharmaceutical Industries
NIOSH: National Institute for occupational safety and health.
OSHA: Occupational health and safety administration.
USP: US Pharmacopeia <800>
Managing hazardous drug exposures: information for healthcare settings.
29CFR: part 1910—Occupational Safety and health standards.
ASHP: American Society of Health-System Pharmacists.