Pharmaceutical fillers play a crucial role in medication effectiveness, stability, and safety. These inactive ingredients help improve drug formulation, ensuring consistent dosage and enhanced absorption. However, not all fillers are created equal—choosing safe, high-quality fillers can significantly affect drug performance and patient health. In this article, we’ll explore the powerful benefits of using safe pharmaceutical fillers and why they are essential in the pharmaceutical industry
In pharmaceutical formulation science, the terms base, carrier, diluent, dry binder, filler, and vehicle are often used interchangeably across many dosage forms. In this general information chapter, the terms will be applied as follows:
- Base: Semisolids (ointments, creams, etc.)
- Carrier: Ordered mixes (solid dosage forms and dry powder inhalation dosage forms)
- Diluent: A General term that may be applied to all other categories
- Dry binder: Solid dosage forms (tablets)
- Filler: Solid dosage forms (capsules, granules, powders, and tablets)
- Vehicle: Liquid and semisolid dosage forms (solutions, suspensions, ointment, creams, etc.)
Pharmaceutical Excipients: Pharmaceutical Filler, Uses, Examples, and Applications
Pharmaceutical excipients are substances other than the active pharmaceutical ingredient (API) that are included in drug formulations. They play a critical role in the development, stability, delivery, and effectiveness of pharmaceutical products. Among the various types of excipients, fillers (also known as diluents) are one of the most commonly used.
Among the most important functional roles fillers play is their ability to impart desirable properties like:
- Manufacturing properties (e.g., powder flow, tablet compaction strength, wet or dry granule formation, or homogeneity)
- Performance (e.g., content uniformity, disintegration, dissolution, tablet integrity, friability, or physical and chemical stability)
- Adjust the overall dosage form weight.
Some fillers (e.g., microcrystalline cellulose) occasionally are referred to as “dry binders” because of the high degree of tablet strength they impart to the final compressed tablet.
This article explores the role of fillers in pharmaceutical formulations, their uses, examples, and applications.
What are pharmaceutical fillers?
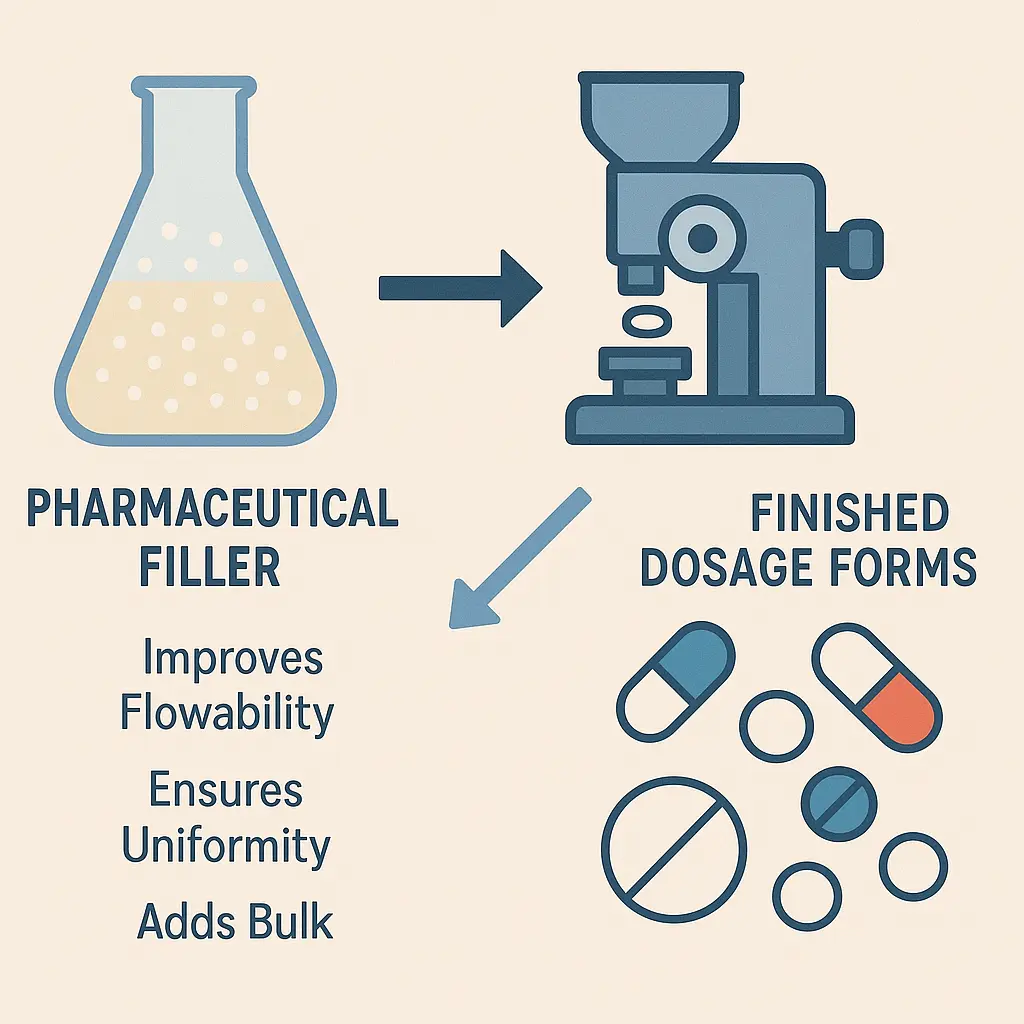
Pharmaceutical fillers are inert substances added to pharmaceutical formulations to increase the bulk volume of a tablet or capsule. They are particularly important when the dosage of the active ingredient is very small, making it difficult to handle or administer. Fillers ensure that the final product has the appropriate size, weight, and consistency for manufacturing, packaging, and patient use.
Pharmaceutical fillers are typically used in solid dosage forms such as tablets, capsules, and powders. They are chosen based on their compatibility with the API, physical and chemical properties, and ability to meet regulatory requirements.
Uses of Fillers in Pharmaceutical Formulations
- Bulk Enhancement: Pharmaceutical fillers add volume to low-dose APIs, making them easier to handle during manufacturing and ensuring accurate dosing.
- Improved Flow Properties: Pharmaceutical fillers enhance the flowability of powders, which is essential for efficient tablet compression and capsule filling.
- Stability: Some Pharmaceutical fillers act as stabilizers, protecting the API from degradation due to moisture, light, or other environmental factors.
- Binders and Disintegrants: Certain fillers also serve as binders (to hold the tablet together) or disintegrants (to help the tablet break down in the body).
- Patient Compliance: Pharmaceutical fillers can improve the taste, texture, and appearance of medications, making them more palatable and easier to swallow.
Examples of Common Pharmaceutical Fillers
- Lactose: Lactose is one of the most widely used fillers in pharmaceutical formulations. It is a naturally occurring sugar derived from milk and is highly compressible, making it ideal for tablet manufacturing. Lactose is compatible with most APIs and is available in various forms, such as anhydrous lactose and lactose monohydrate.
- Microcrystalline Cellulose (MCC): MCC is a purified, partially depolymerized cellulose derived from wood pulp. It is highly compressible and acts as both a filler and a binder. MCC is commonly used in tablets and capsules due to its excellent flow properties and stability.
- Calcium Carbonate: Calcium carbonate is an inorganic filler used in antacid tablets and other formulations. It is cost-effective and provides a source of calcium, making it suitable for nutritional supplements.
- Starch: Starch, derived from corn, potato, or wheat, is a natural filler and disintegrant. It is often used in combination with other excipients to improve tablet disintegration and dissolution.
- Mannitol: Mannitol is a sugar alcohol used as a filler in chewable tablets and orally disintegrating tablets (ODTs). It has a sweet taste and a cooling effect, making it ideal for patient-friendly formulations.
- Dicalcium Phosphate: Dicalcium phosphate is an inorganic filler used in tablets and capsules. It is chemically inert and provides good compressibility and flow properties.
- Sucrose: Sucrose, or table sugar, is used as a filler in lozenges and chewable tablets. It provides sweetness and improves the palatability of medications.
Applications of Fillers in Pharmaceutical Products
- Tablets: Fillers are essential in tablet formulations to ensure proper size, weight, and mechanical strength. They also aid in the uniform distribution of the API.
- Capsules: In capsule formulations, fillers are used to bulk up the powder mixture, ensuring accurate dosing and smooth filling of the capsule shells.
- Powders: Fillers are used in powdered medications to improve flowability and ensure uniform mixing of the API with other excipients.
- Orally Disintegrating Tablets (ODTs): Fillers like mannitol and microcrystalline cellulose are used in ODTs to provide rapid disintegration and a pleasant mouthfeel.
- Chewable Tablets: Fillers such as sucrose and mannitol are used in chewable tablets to improve taste and texture, making them more appealing to patients, especially children.
- Nutritional Supplements: Fillers like calcium carbonate and dicalcium phosphate are used in vitamin and mineral supplements to provide bulk and additional nutritional benefits.
Considerations for Selecting Pharmaceutical Fillers
When selecting a pharmaceutical filler for a pharmaceutical formulation, several factors must be considered:
- Compatibility: The filler must be chemically and physically compatible with the API and other excipients.
- Regulatory Approval: The filler must meet regulatory requirements and be approved for use in pharmaceutical products.
- Cost: The cost of the filler can impact the overall cost of the drug product, so cost-effectiveness is an important consideration.
- Patient Factors: The filler should be chosen with the patient in mind, ensuring that the final product is easy to administer and well-tolerated.
- Manufacturing Process: The filler must be suitable for the intended manufacturing process, whether it is direct compression, wet granulation, or dry granulation.
Conclusion
Pharmaceutical fillers are indispensable components of drug formulations, providing bulk, stability, and improved handling properties. They play a vital role in ensuring the quality, efficacy, and patient acceptability of pharmaceutical products. With a wide range of fillers available, formulators can select the most appropriate excipient based on the drug product’s specific requirements and the patient’s needs. As the pharmaceutical industry continues to evolve, the development of new and improved pharmaceutical fillers will remain a key area of research and innovation.