The study by Maruyama et al. (2025) explores the potential of repeated sequential administration of a pegylated emulsion of SU5416 (PE-SU5416) and liposomal paclitaxel (PL-PTX) in 4T1 triple-negative breast cancer (TNBC)-bearing mice, demonstrating enhanced anti-tumor effects through vascular normalization and tumor microenvironment modulation.[1] While the findings are promising, they invite deeper scrutiny into the mechanisms, translational relevance, and broader implications of this approach. This commentary employs a Socratic framework to dissect the research, identifies challenges in its application, and offers actionable insights for advancing vascular normalization strategies.
Probing Mechanisms and Implications
Why does structural vascular normalization plateau after two PE-SU5416 doses, while functional improvements persist through three?
The study reports that structural normalization—evidenced by pericyte coverage and endothelial integrity—peaked after two PE-SU5416 administrations but declined by the third dose. In contrast, functional metrics like blood flow, oxygen supply, and liposomal distribution improved progressively.[1] This dichotomy suggests that structural changes alone may not fully capture vascular functionality. The transient structural benefits could stem from SU5416’s anti-angiogenic threshold: excessive VEGF inhibition may destabilize nascent vessels, counteracting initial gains.
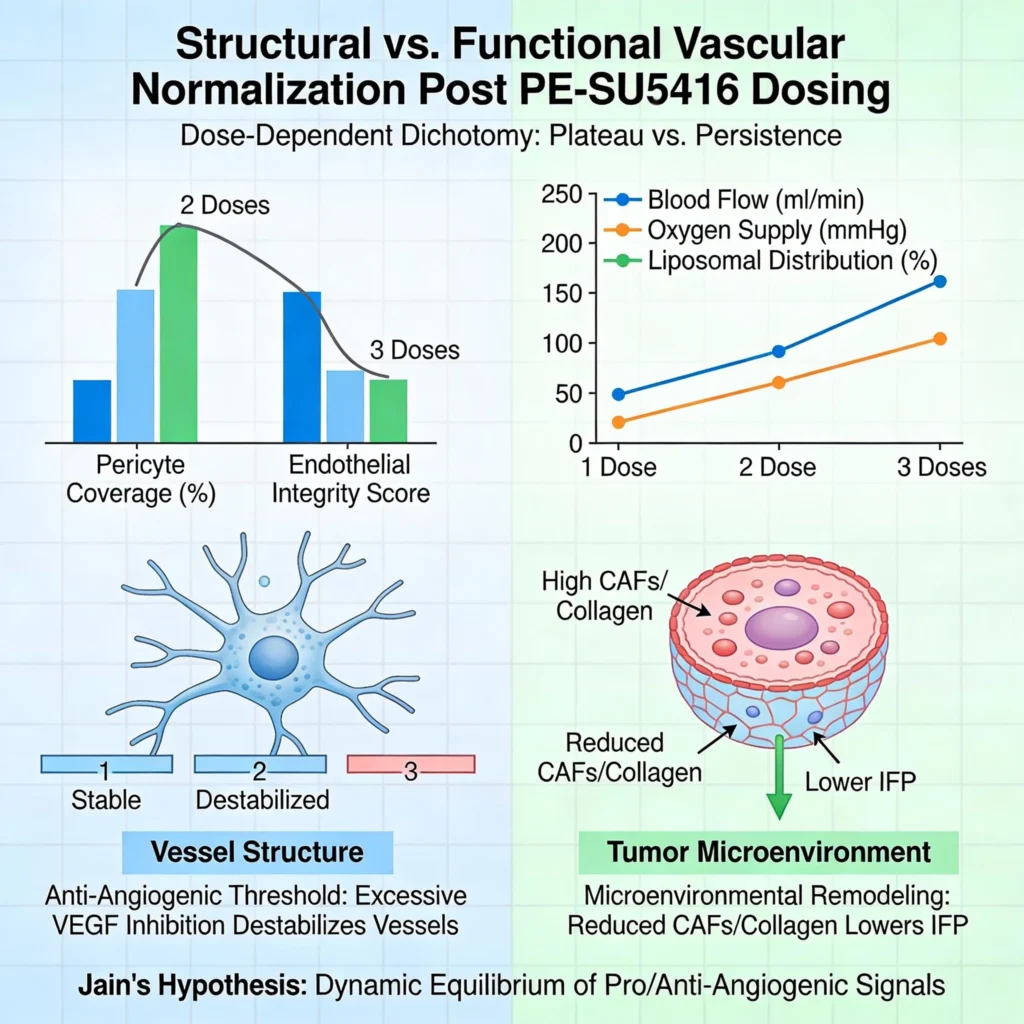
Functionally, reduced cancer-associated fibroblasts (CAFs) and collagen after repeated dosing likely lower interstitial fluid pressure (IFP), enhancing drug penetration independently of vessel architecture. This aligns with Jain’s hypothesis that vascular normalization is a dynamic equilibrium between pro- and anti-angiogenic signals, where functional gains may outlast structural ones due to microenvironmental remodeling.
1. Structural Normalization: A Transient Phenomenon
Structural normalization, characterized by increased pericyte coverage (NG2+ cells) and endothelial integrity (CD31+ cells), peaked after two doses of PE-SU5416 but declined after the third dose [1]. This transient effect aligns with the biphasic nature of anti-angiogenic therapies, where moderate VEGF inhibition stabilizes vessels, but excessive inhibition disrupts endothelial-pericyte interactions, leading to vessel regression.[2] SU5416, as a VEGF receptor-2 kinase inhibitor, likely reaches a threshold beyond which its anti-angiogenic effects become counterproductive. This threshold effect is consistent with prior studies showing that prolonged or high-dose VEGF inhibition can cause “vascular pruning,” reducing vessel density and exacerbating hypoxia.[2] The decline in structural normalization after the third dose may reflect this tipping point, where the balance shifts from vessel stabilization to destabilization.
2. Functional Improvements: Sustained and Progressive
In contrast to structural changes, functional metrics such as blood flow, oxygen supply, and liposomal distribution improved progressively with repeated dosing [1]. This suggests that functional benefits are not solely dependent on vessel architecture but are also influenced by broader changes in the tumor microenvironment. For instance, the reduction in cancer-associated fibroblasts (CAFs) and collagen after repeated PE-SU5416 administration likely lowers interstitial fluid pressure (IFP), a major barrier to drug penetration.[3] Lower IFP enhances convective transport, allowing liposomal drugs like PL-PTX to distribute more evenly within the tumor, even if vessel structure deteriorates. This decoupling of structure and function highlights the importance of considering both metrics when evaluating vascular normalization strategies.
3. Mechanistic Insights: The Role of the Tumor Microenvironment
The dichotomy between structural and functional improvements can be explained by the dynamic interplay between vascular and stromal components of the tumor microenvironment. Jain’s hypothesis posits that vascular normalization is a delicate balance between pro- and anti-angiogenic signals, where functional gains may outlast structural ones due to microenvironmental remodeling.[4] In this study, the reduction in TGF-β levels after repeated PE-SU5416 administration likely suppresses CAF activation, leading to decreased collagen deposition and IFP.[1] This stromal remodeling enhances drug delivery independently of vessel structure, explaining why functional improvements persist even as structural normalization wanes.
4. Implications for Therapeutic Design
The transient nature of structural normalization and the sustained functional improvements have important implications for therapeutic design. First, they suggest that anti-angiogenic therapies should be optimized to avoid excessive VEGF inhibition, which could undermine structural gains. Second, they highlight the need to target stromal components, such as CAFs and ECM, to sustain functional improvements. Combining VEGF inhibitors with stromal-targeting agents (e.g., TGF-β inhibitors or ECM-modulating enzymes) could enhance therapeutic outcomes by addressing both vascular and stromal barriers to drug delivery.[5]
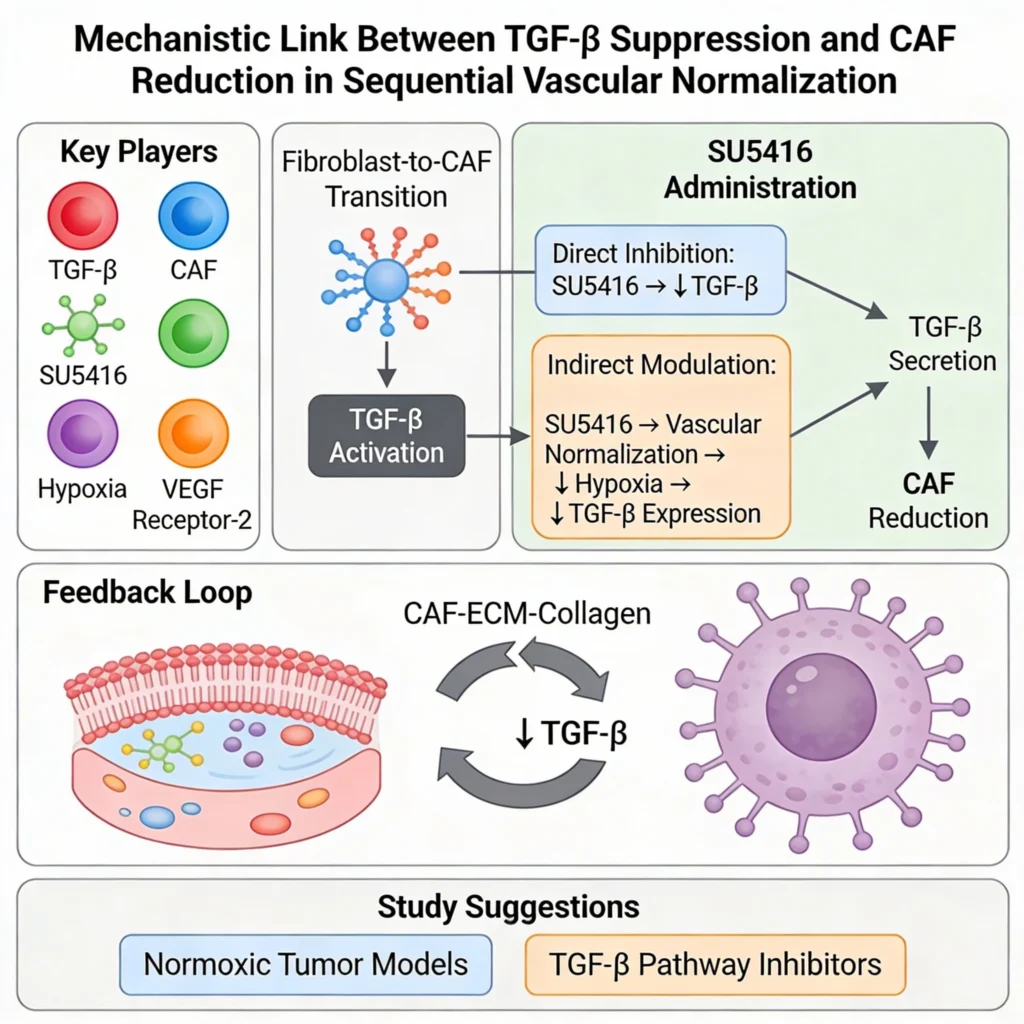
How does TGF-β suppression mechanistically link to CAF reduction?
The authors attribute CAF reduction to decreased TGF-β levels, a key activator of fibroblast-to-CAF transition. However, the precise pathway—whether SU5416 directly inhibits TGF-β secretion or indirectly modulates it via hypoxia alleviation—remains unclear. Hypoxia-driven TGF-β expression is well-documented, and improved oxygenation after vascular normalization could suppress TGF-β, breaking the CAF-ECM-collagen feedback loop. Yet, SU5416’s primary target is VEGF receptor-2, not TGF-β signaling.[6] A follow-up study could isolate hypoxia’s role using normoxic tumor models or TGF-β pathway inhibitors to validate causality.
Why did single PE-SU5416 administration fail to reduce CAFs in this study, contrary to prior work?
Maruyama et al. (2024) previously reported CAF reduction after a single PE-SU5416 dose, but this study observed no significant change until two administrations. Discrepancies may arise from tumor volume differences (100 mm³ vs. 400–500 mm³ at treatment initiation) or microenvironment heterogeneity in larger tumors.[7] Larger tumors may have denser fibrotic regions resistant to short-term therapy, underscoring the need for standardized models in vascular normalization research.
Critical Challenges in Translational Application
1. Sex-Specific Models in Breast Cancer Research
The use of male mice for 4T1 TNBC models, while justified for consistency with prior studies, overlooks sex-specific differences in tumor biology and drug metabolism. Estrogen receptor-negative TNBC disproportionately affects premenopausal women, and murine hormonal environments influence immune responses and metastasis. Future work should validate findings in female models to ensure clinical relevance.
2. Dosing Schedule Translation to Humans
The 48-hour interval between PE-SU5416 and PL-PTX in mice may not directly translate to humans due to pharmacokinetic differences. SU5416’s half-life in humans is shorter (~1.5 hours) than in mice, necessitating adjusted dosing frequencies. Additionally, combining pegylated emulsions with liposomal drugs risks overlapping toxicity profiles, such as hand-foot syndrome from PEGylated formulations. Phase I trials should prioritize pharmacokinetic-pharmacodynamic modeling to optimize schedules.
3. Long-Term Consequences of Vascular Normalization
While repeated PE-SU5416 enhanced drug delivery, prolonged VEGF inhibition risks “vascular pruning,” exacerbating hypoxia and metastasis post-therapy. The study’s 21-day endpoint may miss late-stage rebound effects. Longitudinal tracking of metastasis and survival in resistant clones is critical for evaluating durability.
Actionable Insights and Recommendations
1. Integrate Multimodal Imaging for Comprehensive Vascular Assessment
Structural metrics (e.g., CD31/NG2 co-staining) should be complemented with functional imaging, such as dynamic contrast-enhanced MRI or photoacoustic tomography, to capture real-time perfusion and oxygen gradients. This dual approach would clarify discrepancies between vessel morphology and functionality.
2. Explore Synergy with Immune Checkpoint Inhibitors
CAF reduction and ECM remodeling may enhance T-cell infiltration, potentiating anti-PD-1/PD-L1 therapies. Sequential PE-SU5416, PL-PTX, and immunotherapy could exploit normalized vasculature for improved immune cell access, a strategy gaining traction in TNBC.
3. Address Scalability of Pegylated Formulations
PE-SU5416’s efficacy hinges on nanoparticle stability and tumor accumulation. Batch-to-batch variability in PEGylation efficiency or emulsion size (134.6 nm ± 1.2 nm) could affect reproducibility.[1] Implementing quality-by-design (QbD) principles during manufacturing will ensure consistency in clinical-grade production.
4. Mitigate Pitfalls in Preclinical Models
- Standardize Tumor Volumes: Initiate treatments at uniform sizes (e.g., 100 mm³) to reduce microenvironment variability.
- Incorporate Orthotopic Models: Subcutaneous 4T1 tumors lack the breast tumor’s anatomical context; orthotopic implants better mimic metastatic spread.
- Monitor Off-Target Effects: SU5416’s kinase inhibition may affect non-tumor tissues. Toxicity screens for cardiac, hepatic, and renal impacts are essential.
Conclusion
Maruyama et al. provide compelling evidence that repeated vascular normalization enhances drug delivery and efficacy in aggressive TNBC. However, the interplay between structural and functional vascular changes, alongside TGF-β’s role in CAF modulation, warrants deeper mechanistic exploration. Translational success will depend on addressing sex-specific biology, optimizing dosing for human physiology, and integrating complementary therapies. As vascular normalization evolves from a concept to a clinical tool, interdisciplinary collaboration—bridging drug delivery, oncology, and immunology—will be paramount to overcoming TNBC’s therapeutic challenges.


