Core-Shell Nanocarriers for the Codelivery of Interferons in Long-Acting Drug Delivery Systems
Recent advancements in nanotechnology have catalyzed the development of multilayer nanocarriers for drug delivery, particularly for delivering biotherapeutics such as interferons (IFNs). This technical commentary delves into the study by Ramos et al., which introduces a core-shell nanoparticle system designed for the codelivery of interferon-alpha (IFN-α) and interferon-gamma (IFN-γ) as a long-acting antiviral therapy. By addressing key challenges in stability, bioavailability, and controlled release, this innovative delivery system holds promise in enhancing antiviral efficacy. The commentary evaluates the methodology, materials, physicochemical characterization, and biological insights, providing a critical analysis of the study\’s potential impact on clinical applications.1,2
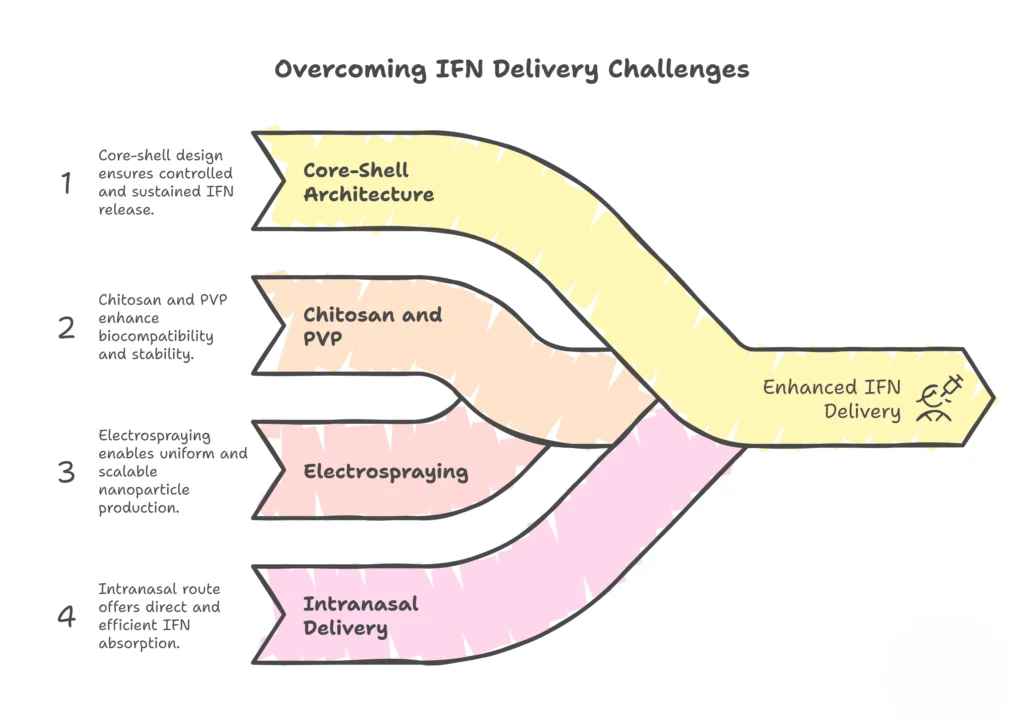
Interferons (IFNs) are pivotal in orchestrating immune responses against viral infections, cancers, and autoimmune diseases. Despite their therapeutic potential, clinical applications of IFNs have been constrained by challenges in pharmacokinetics, rapid clearance, and systemic toxicity. The multilayer nanocarrier developed by Ramos et al. leverages core-shell architecture with chitosan (CS) and polyvinylpyrrolidone (PVP) to encapsulate IFN-α and IFN-γ, providing a controlled release platform that overcomes these limitations. The study emphasizes electrospraying as a robust technique for nanoparticle synthesis, yielding biocompatible and biodegradable particles suitable for intranasal delivery.1,3
The significance of nanocarriers lies in their ability to bypass traditional barriers in drug delivery, such as mucosal membranes, by enhancing permeability and retention. Nasal delivery offers several advantages, including rapid systemic absorption and avoidance of hepatic first-pass metabolism. Thus, the study’s focus on intranasal administration is strategically aligned with the need for efficient antiviral responses, particularly in respiratory tract infections
The methodology employed for synthesizing the core-shell nanoparticles is grounded in coaxial electrospraying, a technique known for producing monodisperse particles with high encapsulation efficiency. The study’s choice of chitosan as the core material is particularly noteworthy due to its mucoadhesive properties and ability to transiently open tight junctions, facilitating enhanced drug absorption across epithelial barriers. PVP, used as the outer shell, contributes to the rapid release of IFNs due to its hydrophilic nature, making it an optimal choice for achieving a biphasic release profile.1
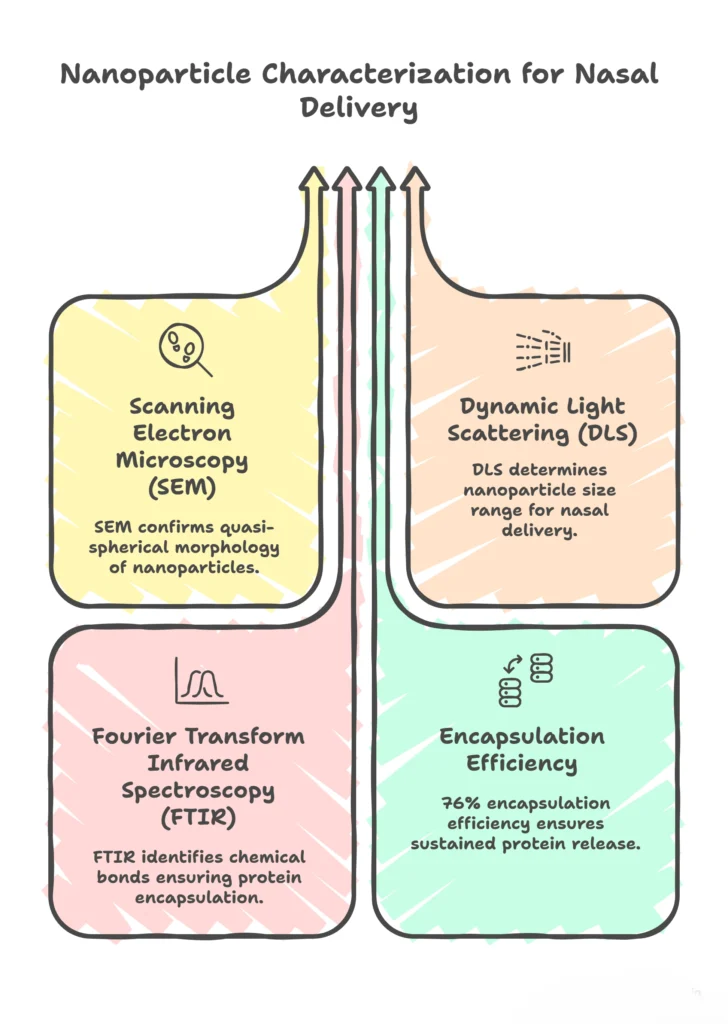
Physicochemical characterization, including Scanning Electron Microscopy (SEM), Dynamic Light Scattering (DLS), and Fourier Transform Infrared Spectroscopy (FTIR), reveals that the nanoparticles exhibit a quasi-spherical morphology with a size range conducive to nasal delivery. The encapsulation efficiency of approximately 76% underscores the effectiveness of the formulation in retaining therapeutic proteins, a critical parameter for ensuring sustained release and bioavailability.
Biocompatible Multilayer Nanocarriers for Controlled Release of Interferons
The biological evaluation of the nanoformulation demonstrates promising results in terms of antiviral efficacy and cytocompatibility. The in vitro release kinetics indicate a controlled, biphasic release pattern, with an initial burst release followed by sustained release over 14 days. This kinetic profile is advantageous for maintaining therapeutic concentrations of IFNs over extended periods, thereby reducing dosing frequency and minimizing systemic toxicity.3,4
Cell viability assays conducted on HeLa and HFF cell lines confirm the formulation’s safety profile, with no significant cytotoxic effects observed at therapeutic concentrations. Additionally, the antiviral activity against Mengo virus, assessed through the inhibition of cytopathic effects, highlights the formulation’s potential in combating respiratory viruses.
While the study provides compelling evidence of the multilayer nanocarrier’s efficacy, several areas warrant further investigation. The long-term stability of the nanoparticles under physiological conditions, particularly in the context of storage and transport, remains a critical consideration. Furthermore, in vivo studies in larger animal models and clinical trials will be essential to validate the formulation’s safety and efficacy in humans. The incorporation of targeting ligands or functional excipients could further enhance the specificity and therapeutic index of the nanocarrier system. Additionally, exploring alternative routes of administration, such as pulmonary delivery via inhalers, may expand the clinical applicability of this technology in treating a broader range of respiratory diseases.
The multilayer nanocarrier system developed by Ramos et al. represents a significant advancement in antiviral drug delivery, offering a biocompatible, long-acting platform for the codelivery of IFNs. By addressing key challenges in stability, bioavailability, and controlled release, this innovative approach has the potential to transform the therapeutic landscape for viral infections and beyond. Continued research and development in this area are poised to unlock new frontiers in precision medicine and targeted therapy.1,3,4
Technical Commentary on Methods and Materials: Elaboration on the Methodology and Physicochemical Insights of Core-Shell Nanoparticles1
The methodology underlying the synthesis of core-shell nanoparticles in the study relies on the coaxial electrospraying technique, a precision-driven process that effectively addresses the challenges of controlled nanoparticle fabrication. Coaxial electrospraying operates by applying a high electric field to a coaxial nozzle system that concurrently delivers two immiscible solutions: one for the core and another for the shell. This dual-fluid system allows the generation of monodisperse particles with distinct core and shell architectures, enabling the encapsulation of bioactive molecules in a controlled and scalable manner.
Unlike conventional encapsulation methods, coaxial electrospraying ensures a high encapsulation efficiency by minimizing protein loss during processing. The simultaneous application of the electric field and solvent evaporation creates solid, dry nanoparticles with a core-shell structure, eliminating the need for additional solvent removal steps. These attributes are critical when dealing with sensitive biomolecules like interferons, which are prone to degradation under harsh processing conditions.
The choice of materials for the nanoparticle components further enhances the system’s functional performance. Chitosan, a natural polysaccharide, is employed as the core material due to its unique physicochemical and biological properties. Its mucoadhesive nature allows the nanoparticles to adhere to the mucosal surfaces of the nasal cavity, facilitating prolonged residence time and intimate contact with epithelial cells. This interaction enhances drug permeation through the transient opening of epithelial tight junctions, a phenomenon attributed to chitosan’s cationic nature and ability to interact with negatively charged cellular membranes. Moreover, chitosan’s biodegradability and biocompatibility make it an ideal carrier for intranasal delivery, ensuring safety and minimizing adverse reactions.
Polyvinylpyrrolidone (PVP), used as the shell material, complements chitosan by enabling rapid release of interferons during the initial phase of delivery. The hydrophilic and highly soluble nature of PVP facilitates the creation of a biphasic release profile. Upon contact with the aqueous environment of the nasal mucosa, the PVP shell dissolves, releasing its payload of interferons in a burst phase. This is followed by the gradual degradation of the chitosan core, which sustains the release of interferons over an extended period. Such a biphasic release pattern is advantageous for maintaining therapeutic levels of the drug in the system while minimizing the need for frequent administration, addressing a critical limitation in interferon therapy.
Physicochemical characterization of the nanoparticles validates the integrity and functionality of the core-shell design. Scanning Electron Microscopy (SEM) provides detailed visualization of the nanoparticles, revealing their quasi-spherical morphology and smooth surface, which are ideal for consistent deposition on mucosal surfaces. Dynamic Light Scattering (DLS) measurements further confirm the size distribution of the nanoparticles, which falls within the nanometric range conducive to nasal delivery. Nanoparticles of this size are small enough to penetrate mucosal barriers yet large enough to avoid rapid clearance from the nasal cavity. Fourier Transform Infrared Spectroscopy (FTIR) analysis ensures that the structural integrity of the interferons and polymers remains intact post-encapsulation, a critical factor in preserving the bioactivity of the therapeutic agents.
The encapsulation efficiency of approximately 76%, as determined by molecular exclusion high-performance liquid chromatography (SEC-HPLC), highlights the success of the electrospraying technique in retaining therapeutic proteins. High encapsulation efficiency is paramount for ensuring the sustained release of bioactive molecules and maintaining their therapeutic efficacy over the dosing period. The effective retention of interferons within the nanoparticle system underscores its potential for delivering a stable and controlled therapeutic dose, particularly for respiratory tract infections where consistent and prolonged exposure to antiviral agents is essential.
Overall, the combination of coaxial electrospraying, chitosan’s biofunctional properties, and PVP’s hydrophilicity represents an innovative and effective approach to overcoming the pharmacokinetic challenges associated with interferon therapies. This methodology not only maximizes drug bioavailability but also minimizes systemic side effects, paving the way for more efficient and patient-friendly antiviral treatments.
Biological Characterization and Therapeutic Implications: Biological Characterization and Therapeutic Implications of Core-Shell Nanoformulations1,5,6
The biological evaluation of the core-shell nanoparticle formulation, encapsulating interferon-alpha (IFN-α) and interferon-gamma (IFN-γ), unveils a significant advancement in therapeutic drug delivery, with noteworthy implications for antiviral therapy. The in vitro release kinetics of the formulation demonstrate a well-controlled, biphasic release pattern. This kinetic behavior begins with an initial burst phase, where a significant fraction of the active ingredients is rapidly released. This phase is attributed to the dissolution of the polyvinylpyrrolidone (PVP) outer shell upon exposure to the aqueous environment of the nasal mucosa.
Following this, the formulation enters a sustained release phase, where the degradation of the chitosan (CS) core regulates the gradual release of the encapsulated interferons. Such a controlled release system ensures prolonged therapeutic concentrations of IFNs, extending over a 14-day period. This extended release minimizes the need for frequent administration, addressing one of the major limitations of conventional interferon therapies, which typically require frequent dosing due to rapid clearance and short half-life. By maintaining steady therapeutic levels, the formulation reduces the risk of systemic toxicity associated with high-dose administration, enhancing patient safety and compliance.
The biphasic release profile is particularly advantageous in the context of antiviral therapy. The initial burst provides a rapid onset of action, critical for establishing early immune responses against acute viral infections. This is followed by the sustained phase, which ensures continued immune activation and viral suppression. This kinetic profile is also expected to align with the pharmacodynamic requirements of interferons, as these cytokines rely on prolonged systemic exposure for maximal efficacy. Furthermore, the prolonged presence of IFNs in the target site not only enhances their antiviral action but also sustains their immunomodulatory roles, fostering adaptive immune responses crucial for long-term protection against viral re-infections.
The cytocompatibility of the nanoformulation was rigorously evaluated through cell viability assays conducted on HeLa (human cervical cancer) and HFF (human foreskin fibroblast) cell lines. These assays revealed that the nanoformulation, even at therapeutic concentrations, did not exert significant cytotoxic effects on either cell line. This finding underscores the biocompatibility of the selected polymers, chitosan and PVP, both of which have been extensively documented for their safety profiles in biomedical applications. The absence of cytotoxicity is particularly noteworthy given the encapsulation of bioactive molecules like interferons, which can exhibit dose-dependent toxicity. The results suggest that the core-shell architecture effectively shields the payload during delivery, releasing it in a controlled manner that avoids localized high concentrations and subsequent cellular damage.
The antiviral efficacy of the formulation was further validated using a Mengo virus model. By assessing the inhibition of cytopathic effects on HEp-2 cells, the study demonstrated the potent antiviral activity of the encapsulated interferons. The observed reduction in cytopathic effects indicates that the formulation successfully delivers bioactive interferons to the site of viral replication, enhancing their antiviral mechanism. This includes the induction of antiviral genes, inhibition of viral protein synthesis, and activation of natural killer cells, all of which are central to the immune-mediated eradication of viral infections. Such results are promising for the treatment of respiratory viruses, which often require rapid and sustained immune activation for effective clearance.
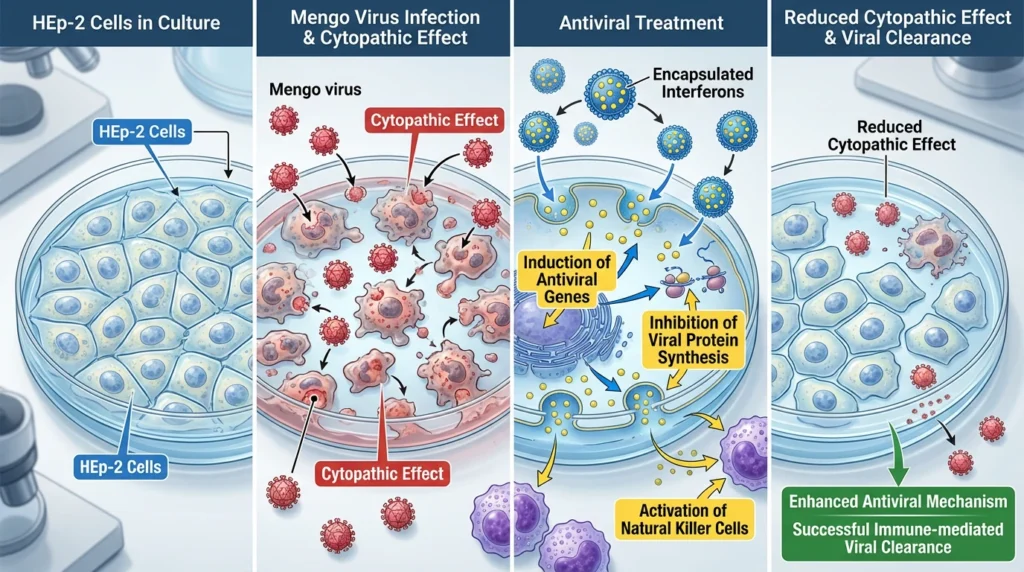
Moreover, the synergistic action of IFN-α and IFN-γ, facilitated by their co-encapsulation, further amplifies the formulation’s therapeutic potential. IFN-α is known for its potent antiviral and antiproliferative effects, while IFN-γ primarily enhances the immunomodulatory pathways, including macrophage activation and antigen presentation. The co-delivery of these cytokines ensures a comprehensive antiviral response that addresses both the innate and adaptive arms of immunity. The therapeutic implications extend beyond antiviral applications, as such formulations could be explored for treating other diseases where interferons play a role, including certain cancers and autoimmune conditions.
In conclusion, the biological characterization of this core-shell nanoparticle formulation highlights its efficacy and safety as a novel therapeutic platform. The biphasic release profile, combined with the absence of cytotoxic effects and robust antiviral activity, positions this formulation as a highly promising candidate for respiratory viral infections and other interferon-mediated therapies. The findings emphasize the potential of nanotechnology-driven drug delivery systems to overcome longstanding challenges in biotherapeutic applications, paving the way for innovative treatments with improved efficacy, safety, and patient compliance.
Future Directions and Perspectives on Multilayer Nanocarriers in Antiviral Therapy1,2,7,8
Although the study by Ramos et al. provides compelling evidence supporting the efficacy of the multilayer nanocarrier system for delivering interferon-alpha (IFN-α) and interferon-gamma (IFN-γ), it opens several avenues for further exploration. A critical area of focus lies in the long-term stability of the nanoparticles under physiological and environmental conditions. Stability concerns are pivotal in ensuring the formulation’s viability for widespread clinical use, especially in contexts where a cold chain is impractical or unavailable.
The nanoparticles’ degradation profiles under varied temperature and humidity conditions need comprehensive evaluation to establish their shelf-life and robustness during transport. Furthermore, encapsulated interferons must retain their bioactivity over prolonged storage periods, as any denaturation could compromise the therapeutic efficacy. Implementing lyophilization or other stabilization techniques during manufacturing may mitigate these risks, but this necessitates additional research to confirm compatibility with the nanoformulation’s physicochemical properties.
Equally critical is the translation of these findings into in vivo studies involving larger animal models. While preliminary evaluations in smaller organisms may provide proof of concept, larger models offer insights into pharmacokinetics, biodistribution, and immune responses that more closely mimic human physiology. Such studies are essential to validate the safety profile, as even biocompatible polymers like chitosan and polyvinylpyrrolidone (PVP) may exhibit unanticipated toxicity or immunogenicity when scaled for clinical dosages.
Moreover, animal studies can elucidate potential off-target effects and establish dosage optimization, particularly for cytokines like interferons that exhibit dose-dependent toxicity. Transitioning from preclinical to clinical trials requires addressing regulatory frameworks, including the FDA’s nanoscale characterization requirements, to ensure the formulation meets stringent safety and efficacy standards before human use.
The incorporation of targeting ligands or functional excipients represents another promising avenue for enhancing the specificity and therapeutic index of the nanocarrier system. Ligands such as peptides, antibodies, or small molecules that bind selectively to overexpressed receptors on viral-infected cells or immune cells could dramatically improve targeted delivery.
By reducing nonspecific uptake, these modifications would enhance therapeutic efficacy while minimizing systemic exposure and associated side effects. Additionally, functional excipients like penetration enhancers or stabilizing agents could be integrated to further improve bioavailability and protect the integrity of the encapsulated interferons. These modifications, however, require meticulous design to ensure compatibility with the core-shell architecture and maintain the biphasic release kinetics critical for sustained therapeutic action.
Exploring alternative routes of administration beyond the intranasal delivery proposed in the study could significantly expand the clinical applicability of this technology. Pulmonary delivery via inhalers is a particularly intriguing option, as it allows for direct drug deposition into the lower respiratory tract, targeting a broader spectrum of respiratory diseases, including severe viral infections like COVID-19 or influenza. Pulmonary administration could provide enhanced absorption efficiency and rapid therapeutic onset due to the large surface area and rich vascularization of the lungs. However, adapting the formulation for pulmonary delivery requires addressing aerosolization efficiency, particle size optimization for deep lung penetration, and ensuring the stability of the nanoparticles under the high shear forces generated during nebulization or inhalation.
The multilayer nanocarrier system developed by Ramos et al. signifies a paradigm shift in antiviral drug delivery. By addressing the challenges of rapid clearance, poor bioavailability, and systemic toxicity, this technology offers a biocompatible, long-acting platform for the codelivery of interferons. Its potential extends beyond antiviral applications to other therapeutic areas, such as cancer immunotherapy or autoimmune disease management, where controlled cytokine delivery is paramount. The innovative core-shell design, coupled with its scalable manufacturing process, aligns well with the demands of modern healthcare for precision medicine solutions.
Continued research and development in this domain are poised to unlock transformative possibilities in targeted therapy. Advancements in nanoparticle engineering, combined with integrative approaches leveraging systems biology and artificial intelligence for drug optimization, could refine these delivery systems further. The horizon for multilayer nanocarriers is expansive, offering the promise of revolutionizing the therapeutic landscape for complex diseases, particularly those requiring intricate immune modulation. With strategic collaboration between researchers, clinicians, and industry stakeholders, this technology holds the potential to redefine treatment paradigms and set new benchmarks in patient care.


