Hazardous drugs refer to substances that can pose significant health risks to individuals, particularly those involved in their manufacturing, handling, or administration. According to guidelines from the National Institute for Occupational Safety and Health (NIOSH), the Occupational Safety and Health Administration (OSHA), and the American Society of Health-System Pharmacists (ASHP).

Drugs are considered hazardous if they meet any of the following criteria:
1. CDER Approved for Human Use
The drug must be approved by the Food and Drug Administration’s Center for Drug Evaluation and Research (FDA-CDER) for human consumption. However, the approval does not necessarily guarantee safety from occupational hazards, especially for healthcare workers exposed to these drugs in workplaces.
2. Regulated by the U.S. Nuclear Regulatory Commission
Drugs don’t fall under the regulation of the U.S. Nuclear Regulatory Commission (NRC), which governs radiopharmaceuticals. Thus, they are regulated differently and are typically not radioactive, but still pose substantial risks.
3. Exhibit Toxicity Criteria in Humans
Hazardous drugs exhibit one or more of the following toxic effects in humans:
- Carcinogenicity: Drugs that have the potential to cause cancer in humans.
- Teratogenicity: Drugs that may cause birth defects in a developing fetus.
- Fertility Problems: Drugs that can impair reproductive health or cause infertility in both men and women.
- Organ Toxicity: Drugs that can damage specific organs (e.g., liver, kidneys) after exposure.
- Genotoxicity: Drugs that can cause genetic damage, such as mutations or chromosomal alterations.
- Similar Structure & Toxicity Profile: Drugs with a similar molecular structure and toxic effects to other known hazardous drugs.
NIOSH Updates Hazardous Drug List to Safeguard Healthcare Workers
Antineoplastic medications, anesthetics, certain antivirals, immunosuppressants, oral contraceptives, and other drugs are recognized as hazardous medications due to their potential health risks. In September 2004, NIOSH (National Institute for Occupational Safety and Health) released an alert aimed at preventing occupational exposure to these hazardous medications in healthcare settings. This alert, titled Preventing Occupational Exposures to Antineoplastic & Other Hazardous Drugs in Healthcare Settings, was published on the CDC website
The 2004 list has been updated multiple times, with major revisions in 2010, 2012, 2014, and 2016. In the 2016 update, 34 new drugs were added to the list, including newly FDA-approved drugs and those with new safety warnings.
In May 2020, NIOSH released a draft update, which further refined the list of hazardous medications, providing an important resource for healthcare professionals to ensure safety. The most recent draft is available NIOSH Hazardous Drugs List 2020
NIOSH follows a sequential methodology enabling the assessment and interpretation of scientific data to find out whether an FDA-approved drug conforms to the NIOSH categorization of hazardous medications. The drugs or molecules in the list are classified as “known to be a human carcinogen”, not classified as “known to be a human carcinogen”, or classified as causing “reproductive hazard”.
At-Risk Workers: Who Faces Health Hazards from Hazardous Drugs in the Workplace?
A wide range of professionals are at risk of health hazards due to exposure to hazardous drugs in the workplace. These include:

- Laboratory Personnel: Handling and testing potentially dangerous substances.
- Nurses: Administering chemotherapy, immunosuppressants, and other hazardous medications.
- Physicians: Prescribing and working with hazardous medications in patient care.
- Pharmacists: Dispensing hazardous medications and managing prescriptions for patients.
- Community Health Workers: Providing care in environments where hazardous medications may be present.
- Housekeeping Staff: Cleaning areas contaminated by hazardous drug residues.
- Shipping, Receiving, and Transport Services Personnel: Handling and transporting hazardous medications safely.
- Healthcare Assistants: Assisting with patient care and drug administration in potentially hazardous environments.
- Personnel at Hazardous Drug Manufacturing Sites: Involved in the production and packaging of hazardous medications.
These workers may be exposed to hazardous drugs during handling, administration, transport, or cleanup, making it crucial for healthcare organizations to implement strict safety protocols to minimize risk.
”Ensuring Safety”: The Importance of Competent Training for Handling Hazardous Drugs
All personnel handling hazardous drugs should be qualified and well-trained to be competent for the development and implementation of suitable, purposeful SOPs. Competent personnel in charge should be well aware of and understand all the necessary practices and precautions. Also, they should regularly evaluate the implemented procedures to prevent health harm to patients, lessen or prevent contamination, and minimize the risk of exposure.
Primary Responsibilities of Competent Personnel
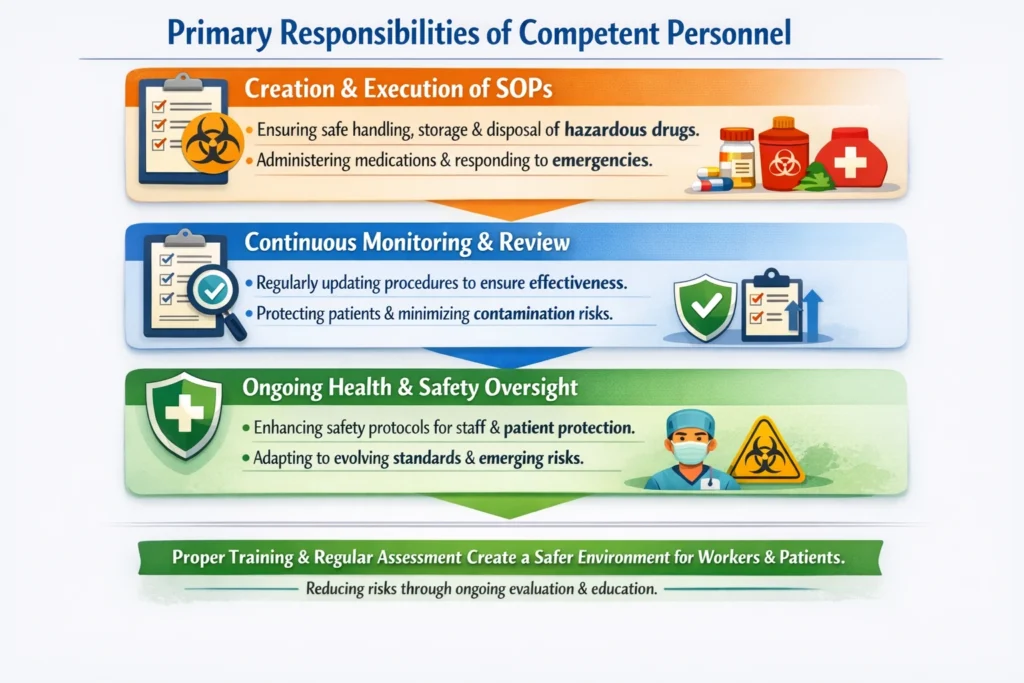
*Creation and Execution of SOPs: Making sure that the necessary procedures are in place for the safe handling, storage, and disposal of hazardous-drugs, including administering medications, responding to emergencies, and managing waste safely.
*Continuous Monitoring and Review: Regularly assessing and updating the procedures to ensure they remain effective. This process helps protect patients, reduce the risk of contamination, and limit exposure to harmful substances.
*Ongoing Health and Safety Oversight: Continuously evaluating and enhancing safety protocols to safeguard both patients and healthcare workers from exposure to hazardous-drugs. This includes keeping procedures aligned with evolving safety standards and emerging risks.
By ensuring that personnel are thoroughly trained and that safety protocols are routinely evaluated, healthcare institutions can significantly reduce the dangers associated with handling hazardous drugs. In the long run, prioritizing proper training and regular assessment creates a safer environment for both workers and patients.
Work Activities with High Risk of Exposure to Hazardous Drugs
Work activities that may have an impact and introduce a high risk of exposure include one or more of the following activities:
- Compounding (sterile or non-sterile)
- Administering hazardous-drugs
- Storage of hazardous-drugs
- Dispensing of medications
- Repackaging hazardous-drugs
- Handling and discarding drug waste
- Decontaminating and deactivating equipment or devices used with hazardous drugs
- Cleaning up spills or exposed surfaces contaminated with hazardous drugs
Importance of Training for Personnel Handling Hazardous Drugs
Personnel who are in charge of handling hazardous products should be well-trained before they independently handle hazardous-drugs. The training program is based on their job functions, such as compounding, receiving, storage, dispensing, and more

The training should cover the following key topics:
- Overview of Hazardous Drugs: A detailed list of active pharmaceutical ingredients (APIs) that are classified as hazardous drugs, along with the associated risks for each.
- Proper Use of Equipment and Devices: How to safely operate and maintain equipment used in handling hazardous-drugs.
- Management of High-Risk Spills: Procedures for handling and containing hazardous drug spills or contamination events.
- Response to Suspected Exposure: Steps to take if there is a suspected exposure to hazardous drugs, including the identification and reporting of symptoms.
- Personal Protective Equipment (PPE): Proper use of gowns, gloves, goggles, face masks, face shields, head covers, shoe covers, and respiratory protection, with clear instructions on when and how to use each type of PPE.
- Proper Disposal of Hazardous Drugs: Guidelines for safely discarding hazardous drugs and residues found on exposed surfaces to prevent contamination.
For more data: USP General Chapter <800> Hazardous Drugs—Handling in Healthcare Settings
References
https://www.cdc.gov/niosh/docs/2020-114/default.html
Safe work practices for handling hazardous drugs
http://dev.uspnf.com/sites/default/files/usp_pdf/EN/USPNF/revisions/800-rb-notice.pdf