A Gateway to Revolutionizing Drug Delivery Systems
Discovered in 1985, fullerenes are a family of carbon-based nanostructures with spherical, ellipsoidal, or tubular geometries, featuring a lattice of sp²-hybridized carbon atoms. Their distinctive arrangement into hexagonal and pentagonal patterns forms a hollow, cage-like structure, with C₆₀ (buckminsterfullerene) being the most iconic example. Resembling a soccer ball, C₆₀ consists of 60 carbon atoms arranged into 20 hexagons and 12 pentagons, offering extraordinary stability, symmetry, and a capacity for electron conjugation. These unique features enable fullerenes to interact with diverse biomolecules, positioning them as promising candidates in drug delivery applications. Despite their potential, fullerenes face challenges such as hydrophobicity and limited water solubility, which necessitate chemical modifications for practical biomedical use.1
Fullerenes exhibit remarkable thermal stability, photochemical activity, and a high surface-to-volume ratio, making them ideal for carrying therapeutic molecules. Their hydrophobic core facilitates the encapsulation of hydrophobic drugs, while chemical functionalization can introduce hydrophilic groups to improve solubility and compatibility with biological systems. For example, hydroxylation produces fullerenols, which display enhanced water solubility and antioxidative properties. Additionally, fullerenes’ electron-donating and accepting capabilities enable redox interactions that are beneficial for targeting oxidative stress-related pathologies. The poor aqueous solubility of pristine fullerenes limits their direct application in biological environments.
To address this, researchers have developed a variety of functionalized derivatives. Covalent modifications, such as attaching carboxylic acids, amino groups, or polyethylene glycol (PEG), enhance their hydrophilicity and reduce potential toxicity. Non-covalent methods, including encapsulation in cyclodextrins or micelles, provide an alternative strategy for improving their biomedical applicability without altering their core structure. These derivatives have demonstrated effectiveness in stabilizing drugs, increasing their bioavailability, and enabling controlled release, especially in cancer therapy and gene delivery systems. 2,3
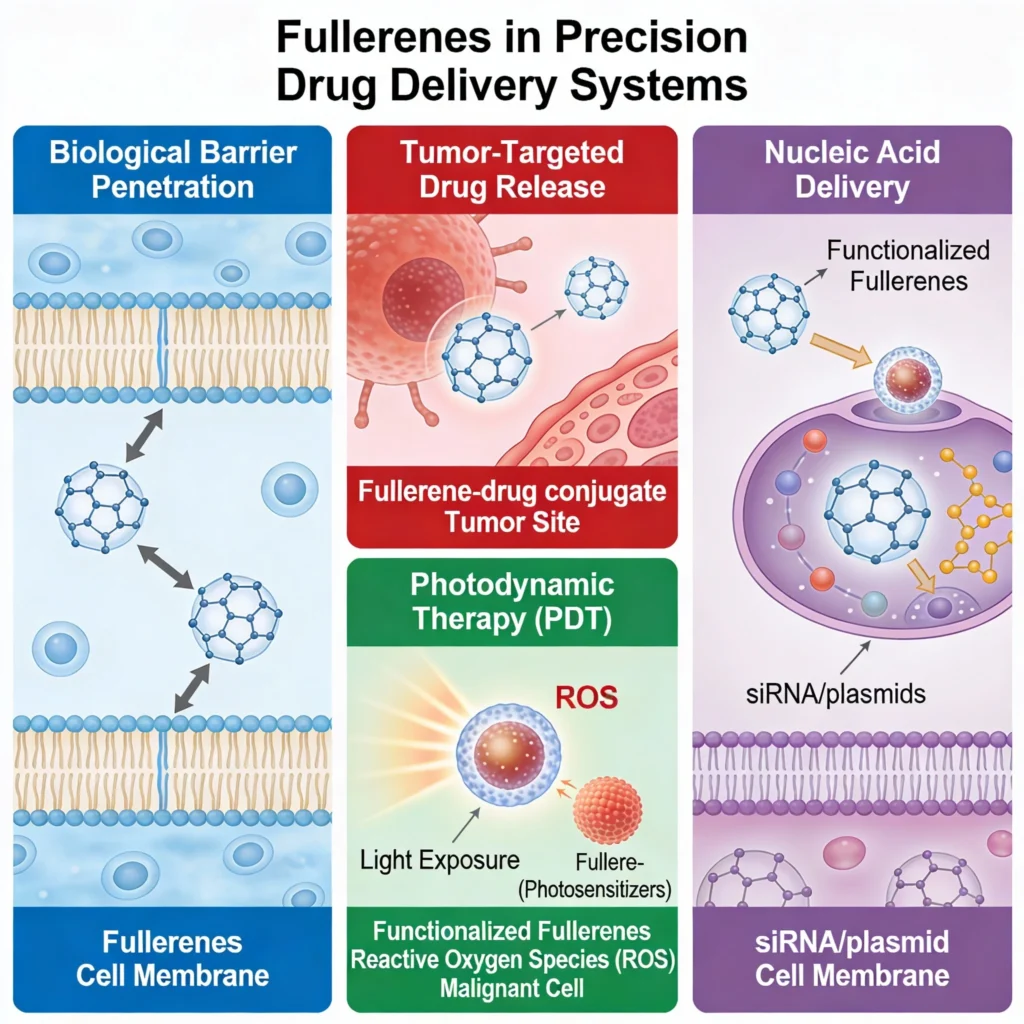
Fullerenes offer unparalleled potential in drug delivery due to their ability to cross biological barriers and deliver therapeutic agents to targeted sites. In cancer treatment, fullerene-drug conjugates have shown the capability to selectively release chemotherapeutics at tumor sites, reducing systemic toxicity. Studies have highlighted their use in photodynamic therapy (PDT), where functionalized fullerenes act as photosensitizers.
When exposed to light, they generate reactive oxygen species (ROS), inducing localized oxidative stress that can destroy malignant cells. Moreover, fullerenes have been successfully employed in nucleic acid delivery, where functionalized derivatives facilitate the transport of siRNA or plasmids across cell membranes without eliciting significant immune responses. 3
Drug Delivery with Nanomaterials and Fullerenes3–5
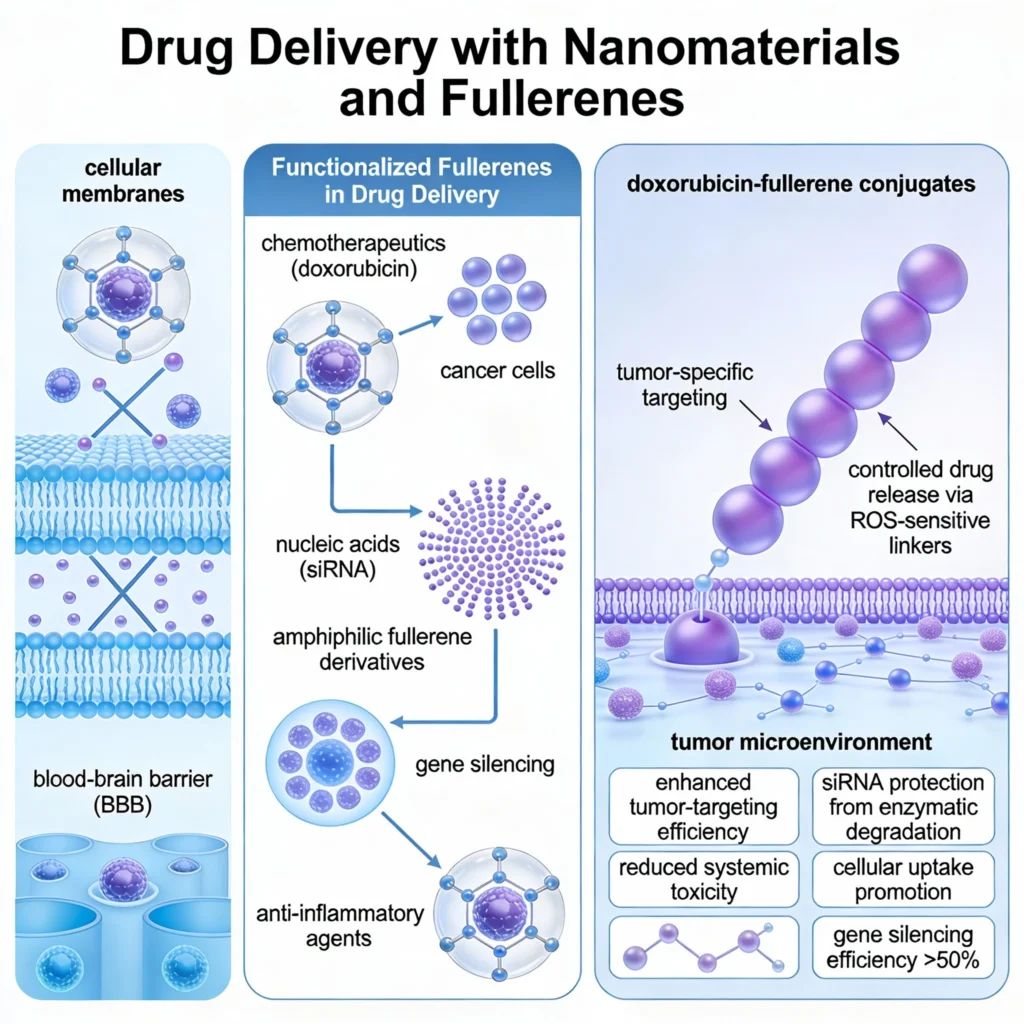
Fullerenes’ unique ability to interact with cellular membranes and biological barriers, such as the blood-brain barrier (BBB), positions them at the forefront of nanotechnology-driven drug delivery systems. Functionalized fullerenes have been explored for their potential to deliver chemotherapeutics, nucleic acids, and anti-inflammatory agents. Studies on doxorubicin-fullerene conjugates, for instance, have shown enhanced tumor-targeting efficiency with reduced systemic toxicity.
This is achieved by leveraging the fullerene scaffold to deliver drugs selectively to cancer cells, reducing off-target effects commonly observed in conventional chemotherapy. In the realm of nucleic acid therapeutics, fullerene derivatives have been employed as carriers for siRNA, enabling precise gene silencing. Recent experiments combining dry lab simulations with wet lab validation have revealed that amphiphilic fullerene derivatives can protect siRNA from enzymatic degradation while promoting cellular uptake. Furthermore, these conjugates show promise in regulating gene expression associated with neurodegenerative and inflammatory diseases, achieving silencing efficiencies exceeding 50% in preclinical models.
In oncology, fullerene-drug conjugates, particularly with doxorubicin, demonstrate high tumor specificity and controlled drug release. Functionalization with reactive oxygen species (ROS)-sensitive linkers allows precise release in tumor microenvironments, where oxidative stress is elevated. This dual mechanism—ROS-mediated drug release and fullerene-induced oxidative cytotoxicity—has proven effective in preclinical models, reducing the adverse effects associated with traditional chemotherapy.
Fullerenes have also emerged as robust carriers for nucleic acids such as siRNA, overcoming barriers like enzymatic degradation and poor cellular uptake. Studies report that amphiphilic fullerene derivatives improve the stability of siRNA and enhance delivery efficiency. Preclinical evaluations reveal successful gene silencing in neurodegenerative and inflammatory disease models, with functionalized fullerenes achieving up to 50% gene regulation efficacy. Computational models further optimize these systems for clinical translation by simulating molecular interactions and stability.
Overcoming the hydrophobicity of pristine fullerenes is critical for biomedical applications. Functionalization strategies, such as attaching hydrophilic polymers or incorporating stimuli-responsive linkers, enhance their solubility and biocompatibility. For instance, ROS-sensitive fullerene systems achieve precise drug release under oxidative conditions. Similarly, PEGylation improves circulation times, enabling targeted delivery. These advances in chemical engineering broaden their therapeutic utility and mitigate potential side effects.
Technical Perspectives: Dry Lab and Wet Lab Insights into Fullerene-Biological Membrane Interactions6–10
Fullerenes, as a novel class of carbon-based nanomaterials, have gained significant attention due to their unique properties, including their ability to interact with biological membranes and serve as potential carriers for drug delivery systems. The mechanistic understanding of how fullerenes interact with cellular environments is multifaceted, and research in both computational (dry lab) and experimental (wet lab) domains has provided crucial insights into their behavior in biological systems.
Dry lab approaches, notably molecular docking and molecular dynamics (MD) simulations, have been instrumental in predicting the interactions between fullerenes and biological membranes. These computational methods allow for the evaluation of fullerenes’ binding affinity to membrane components, such as phospholipids, and the determination of their potential to integrate into or disrupt lipid bilayers. Molecular docking studies have suggested that fullerene derivatives can exhibit strong binding affinities for lipid head groups, thereby influencing membrane fluidity and permeability. In parallel, MD simulations help in understanding the conformational changes of fullerenes upon membrane interaction, providing insights into their transport mechanisms and stability within physiological conditions.
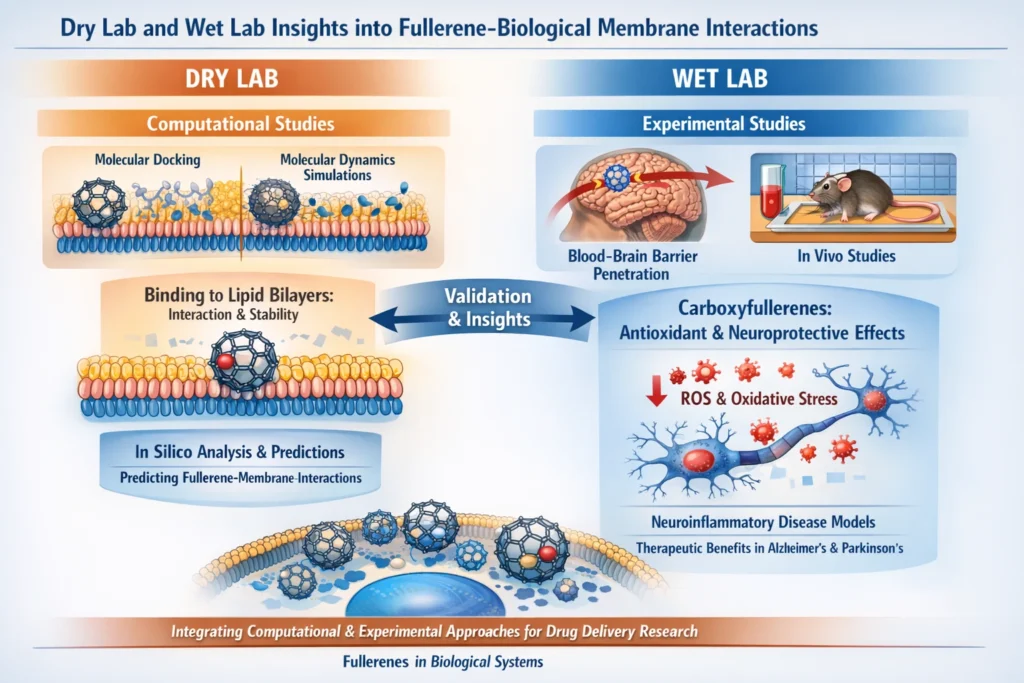
These simulations, which model the dynamic behavior of molecules over time, are crucial for predicting how fullerenes might interact with complex biomolecular systems, including cellular membranes, without the need for experimental validation at early stages. Through these computational tools, researchers can predict how fullerenes will behave under different conditions, thus paving the way for targeted drug delivery applications.
On the experimental front, wet lab studies have continuously provided critical validation for the theoretical predictions made by dry lab techniques. In vitro and in vivo experiments have demonstrated the capability of fullerenes to penetrate biological barriers, such as the blood-brain barrier (BBB), and facilitate targeted drug delivery. Carboxyfullerene derivatives, in particular, have been investigated for their potential in treating neuroinflammatory diseases, where they exhibit potent antioxidant properties.
These derivatives have been shown to effectively scavenge reactive oxygen species (ROS) that are implicated in neurodegenerative diseases such as Alzheimer’s and Parkinson’s disease. Preclinical trials using animal models have shown that these fullerenes not only reduce oxidative stress but also restore neuronal function, demonstrating their therapeutic potential in treating conditions associated with inflammation and oxidative damage in the brain.
Furthermore, fullerenes’ ability to modulate neuroinflammatory pathways and promote neuronal regeneration has been evaluated in vivo, with promising results highlighting their potential as drug delivery carriers capable of improving the bioavailability of therapeutic agents at the targeted site. The combination of dry lab and wet lab findings has thus led to an enhanced understanding of fullerene-based nanomaterials, pushing forward their application in biomedical fields, particularly in neuropharmacology and nanomedicine.
The integration of dry and wet lab approaches offers a comprehensive framework for exploring the full potential of fullerenes in drug delivery applications. Computational models not only predict the interactions of fullerenes with biomolecules but also serve as a guiding tool for optimizing their design for specific therapeutic purposes. Experimental validation in biological systems, on the other hand, ensures that these predictions hold true under realistic conditions.
By combining these two perspectives, researchers can gain a more accurate and holistic understanding of fullerene functionality, ultimately leading to the development of more effective and targeted drug delivery systems. Moreover, as the field of nanomedicine continues to evolve, the collaboration between computational and experimental research will play an increasingly important role in designing next-generation nanotherapeutics with higher precision and lower toxicity, contributing to the growing arsenal of treatments for complex diseases, particularly those involving oxidative stress and inflammation.
Future Prospects in Drug Delivery System Engineering
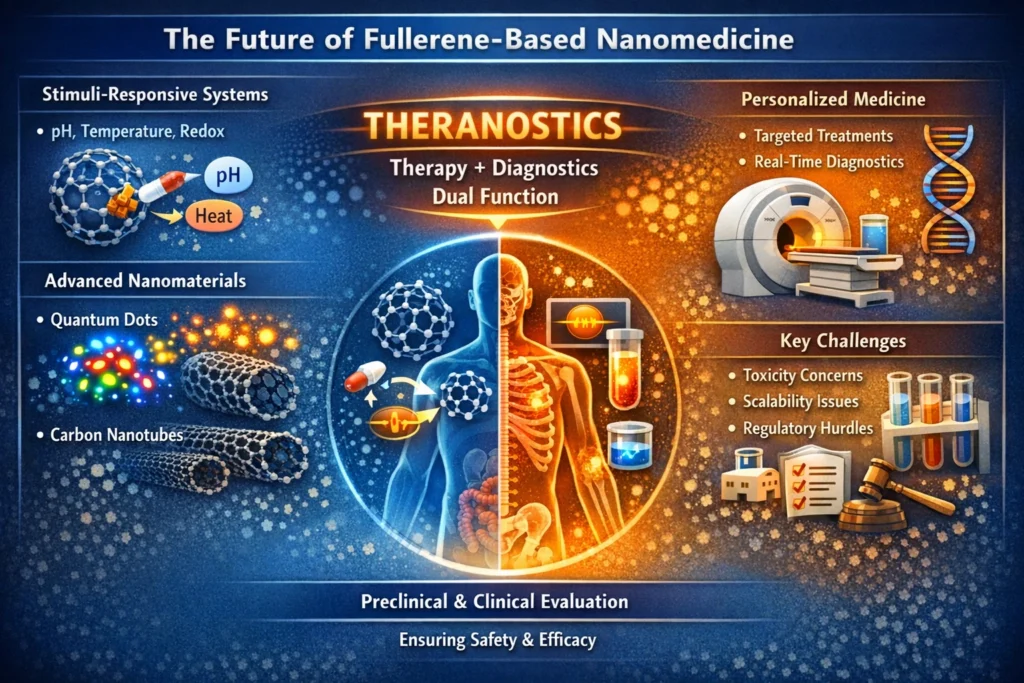
Despite the promising potential of fullerenes in nanomedicine, their clinical application faces several challenges, including concerns about toxicity, scalability, and biodegradability. To overcome these obstacles, researchers are focused on developing biodegradable fullerene derivatives and incorporating them into multifunctional systems, such as theranostic platforms that combine both imaging and therapeutic capabilities.
Interdisciplinary research plays a pivotal role in addressing these concerns, ensuring that fullerene-based therapies are safe, effective, and suited for personalized medicine applications. Looking forward, the future of fullerene-based nanomedicine hinges on the development of intelligent, stimuli-responsive systems that enable precise drug release in response to specific physiological triggers, such as pH, temperature, or redox changes. Integrating fullerenes with other advanced nanomaterials, such as quantum dots and carbon nanotubes, could further enhance their functionality, expanding the scope of their therapeutic applications.
Moreover, the potential of fullerenes as dual therapeutic and diagnostic agents—known as theranostics—presents exciting opportunities in personalized medicine, where tailored treatments can be guided by real-time diagnostic information. However, significant challenges remain on the path to clinical translation, particularly with regard to long-term toxicity, production scalability, and regulatory approval. Overcoming these hurdles will require rigorous interdisciplinary research, coupled with comprehensive preclinical and clinical evaluations to establish the safety and efficacy of fullerene-based systems for widespread biomedical use.
In this context, fullerenes represent a major advancement in nanotechnology with transformative implications for drug delivery. By leveraging their unique structural and chemical properties, researchers are creating innovative drug delivery platforms capable of overcoming the limitations of traditional therapeutic methods. The continued integration of computational modeling, material engineering, and biomedical research will undoubtedly drive the progression of fullerene-based systems into clinical applications, ushering in a new era of targeted, efficient therapeutics.
References
1. Discovery of Fullerenes. www.acs.org/landmarks.


