Lipinski’s Rule or Rule of Five was proposed in 1997 by Christopher A. Lipinski, a medicinal scientist at Pfizer, a set of guidelines designed to assess the drug-likeness of chemical compounds. These principles have played a pivotal role in pharmaceutical research and drug development by helping scientists identify potential drug candidates that are likely to be effective when administered orally. The rule is based on an analysis of the physicochemical properties of known drugs and their absorption characteristics, offering valuable insights into the bioavailability of new compounds.

Lipinski’s Rule of Five consists of a series of criteria that are crucial in determining whether a compound will be successfully absorbed, distributed, metabolized, and excreted by the body — collectively known as ADMET (Absorption, Distribution, Metabolism, Excretion, and Toxicity). It is important to note that Lipinski’s Rule of Five does not assess the drug’s efficacy or pharmacodynamics (the drug’s effect on the body), but focuses instead on the pharmacokinetics, which determine how the body processes the compound after it is administered.
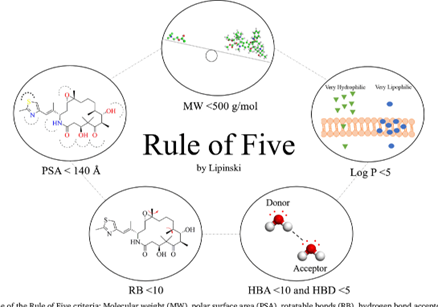
The rule aims to predict oral bioavailability, or the ability of a drug to reach systemic circulation in sufficient quantities when taken by mouth. The five key parameters outlined by Lipinski are molecular weight, lipophilicity (log P), hydrogen bond donors, hydrogen bond acceptors, and rotatable bonds. Each of these factors influences the compound’s ability to traverse biological membranes, particularly those of the gut, and its eventual distribution and effectiveness in the body.
Molecular Weight
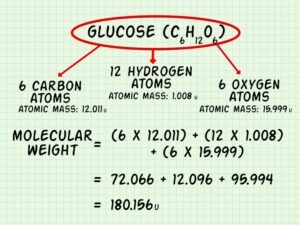
One of the primary criteria in Lipinski’s Rule is that the molecular weight of a compound should be less than 500 Daltons. This size limit helps to ensure that the molecule is small enough to be absorbed effectively across biological membranes. Larger molecules may have difficulty crossing these barriers, resulting in poor bioavailability. This threshold acts as a practical guide to identifying compounds that have a better chance of being effective when administered orally.
Lipophilicity (Log P)
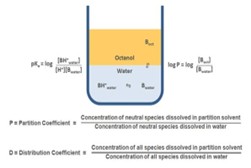
Lipophilicity refers to the compound’s affinity for lipid (fat) environments, and is measured by the partition coefficient, log P. The ideal log P value for optimal absorption is less than 5. This value reflects the balance between the hydrophilic (water-loving) and lipophilic (fat-loving) properties of the molecule. A compound that is too lipophilic may have poor solubility in water, which could hinder its absorption, while a compound that is too hydrophilic may not be able to cross lipid membranes effectively. Therefore, maintaining a log P of less than 5 enhances the compound’s ability to dissolve in both aqueous and lipid environments, improving its absorption and bioavailability.
Hydrogen Bond Donors
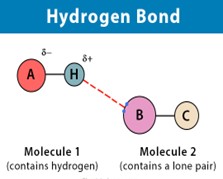
Hydrogen bond donors are functional groups containing nitrogen or oxygen atoms that can form hydrogen bonds with other molecules. Lipinski’s Rule specifies that the number of hydrogen bond donors should be no more than 5. Excessive hydrogen bond donors can make it more difficult for the compound to pass through biological membranes, as they increase the compound’s interaction with water and other solutes. By limiting hydrogen bond donors, the rule ensures that the compound remains stable while maintaining an optimal balance between solubility and permeability.
Hydrogen Bond Acceptors
Alongside hydrogen bond donors, the number of hydrogen bond acceptors (oxygen or nitrogen atoms capable of accepting hydrogen bonds) should not exceed 10. This criterion is important for predicting the solubility and permeability of the compound. A high number of acceptors can lead to excessive interactions with water molecules, reducing the compound’s ability to permeate lipid membranes. Limiting hydrogen bond acceptors enhances the compound’s pharmacokinetic properties, particularly its ability to cross biological barriers.
Rotatable Bonds (Veber’s Modification)
In addition to the core four criteria, Lipinski’s Rule includes a secondary criterion introduced by Veber. This criterion addresses the flexibility of the molecule. Specifically, the number of rotatable bonds — single, non-ring bonds attached to non-terminal, non-hydrogen atoms — should ideally be fewer than 10. Excessive flexibility in a compound can complicate its binding interactions with target proteins and affect its stability. Limiting rotatable bonds helps maintain the molecule’s conformational rigidity, improving its pharmacokinetic properties and reducing the potential for unwanted side effects.
Exceptions to the Rule
While Lipinski’s Rule of Five provides valuable guidance, there are notable exceptions. Some compounds that do not adhere strictly to the rule still exhibit good bioavailability and therapeutic effects. For instance, certain antibiotics, antifungals, vitamins, and cardiac glycosides, which may have larger molecular weights or higher numbers of hydrogen bond donors, can still be effective due to their ability to act as substrates for naturally occurring transporters in the body. These transporters facilitate the absorption of these compounds, allowing them to bypass some of the limitations imposed by Lipinski’s criteria.
Applications in Drug Discovery
Lipinski’s Rule of Five is particularly useful in the early stages of drug discovery and development. During this phase, researchers often have to evaluate large numbers of compounds to determine which ones are most likely to succeed as viable oral medications. By filtering out compounds that do not meet the criteria of Lipinski’s Rule, scientists can focus their efforts on those with a higher likelihood of success, improving efficiency and reducing costs.
In addition to traditional oral bioavailability, Lipinski’s Rule can be applied to compounds targeting the central nervous system (CNS), where stricter criteria are required. For CNS drugs, molecular weight should be less than 300 Daltons, the number of hydrogen bond donors and acceptors should be minimized (fewer than three of each), and the clogP value (a measure of lipophilicity) should be less than 3. These additional constraints help ensure that compounds can cross the blood-brain barrier and reach their target sites in the brain.
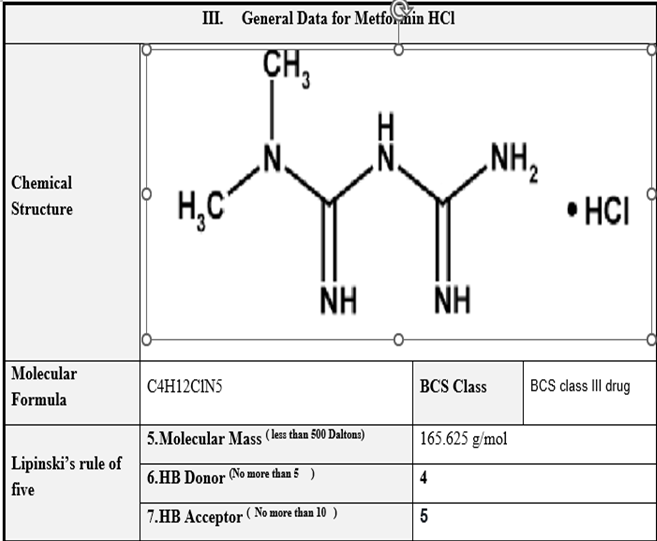
| Hydrogen Acceptor Count | 2 |
| Hydrogen Donor Count | 0 |
| Log P | 2.63 |
| Rotatable Bond Count | 1 |
Conclusion
Lipinski’s Rule of Five remains a foundational tool in medicinal chemistry and drug development. By providing a set of guidelines for assessing the oral bioavailability of compounds, the rule helps streamline the drug discovery process and improve the chances of success for new medications. Despite its limitations, particularly with regard to certain drug classes and compounds with unique pharmacokinetic properties, Lipinski’s Rule continues to be an invaluable resource for researchers seeking to design effective, bioavailable drugs. Understanding and applying these principles can significantly enhance the overall success rate in drug development, ultimately contributing to the creation of safer and more effective therapeutic agents.
References
Lipinski, C. A., Lombardo, F., Dominy, B. W., & Feeney, P. J. (1997). Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Advanced Drug Delivery Reviews, 23(1-3), 3-25.
Wager, T. T., et al. (2016). The Role of Lipinski’s Rule of Five in Drug Discovery. Journal of Medicinal Chemistry, 59(12), 5567-5584.
Duffy, J. (2003). Lipinski’s Rule of Five: A Practical Approach in Drug Development. Pharmaceutical Research, 20(6), 859-870.
Chemaia Naisse sis, Vol 3, Issue 1, POPULAR SCIENTIFIC ARTICLE, 171-177