Drug repurposing is a name given to a strategy for identifying new therapeutic uses for already marketed or FDA-approved drugs that are not related to the original intended medical indications. In other words, drug repurposing is the search for new indications for compounds that have shown an acceptable safety profile in previous clinical trials. Drug repurposing can also be called drug repositioning, drug re-tasking, drug rescuing, drug recycling, drug reprofiling, or therapeutic switching. Moreover, drug repurposing involves the review of abandoned drugs or drugs under investigation that initially showed poor efficacy for another pharmacological indication or have secured a regulatory approval. Pharmaceutical companies are attempting drug repurposing projects for some rare, oncology, infectious, and autoimmune diseases.
Traditional drug discovery is known for being time-consuming, very tough, having a high investment or cost, and having a higher risk of failure. Also, the clinical efficacy and safety profile should be evaluated. On the contrary, the novel approach of drug repurposing has many advantages, such as less time-consuming, less investment cost, less risk of failure, where the clinical efficacy and safety profiles are already available. The risk of failure is lower as the repurposed drug is known to be safe in preclinical studies. The time frame for the drug development is reduced as the preclinical testing, safety assessment, and formulation establishment might already be completed.
Historically, the most successful examples of such repurposing strategy are the repurposing of sildenafil citrate for erectile dysfunction and the repurposing of thalidomide for erythema nodosum leprosum.
Approaches to drug repurposing
Generally, Pharmaceutical entities follow one of the following approaches to drug repurposing as part of a systemic strategy, namely, drug-centric, disease-centric, and target-centric
It should be noted that the nature of the huge data from such mentioned projects and the use of high-throughput technologies pose marked challenges for analysis and effective application both in new drug discovery and drug repurposing. Post-marketing surveillance data and clinical trial data are significantly important sources of data, but their access may be restricted for commercial or confidentiality reasons. However, it is well realized that revealing such wealth of information can aid in further drug development research.
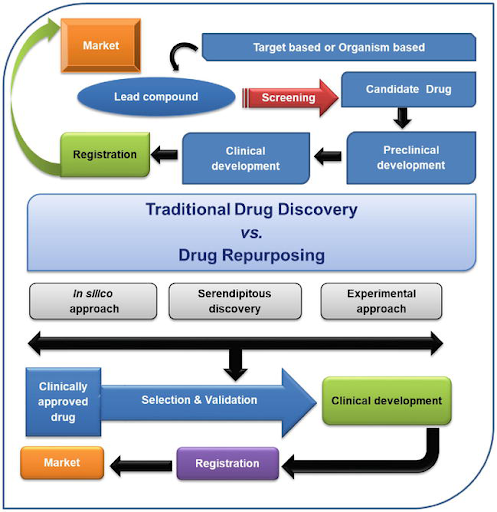
Figure 1: Traditional drug discovery vs drug repurposing
| Drug-oriented information on drugs | Disease-oriented | Target (treatment)-oriented |
|---|---|---|
-Discovering off-label use of approved drugs -Phenotype screening (identifying compounds that show disease-relevant effects) -Target 3D structure of the drug -Chemical structure of drugs and ligands -An adverse effect of drugs | -Information on disease pathway ( constructing disease networks based on gene expression patterns, disease pathology) -Disease omics data( homologous underlying biological mechanisms) -Genetic data of disease (genotyping technology) -Protein interaction network | -Disease omics data -All information related to treatment strategies -Genetics genomics ( immortalized human cancer cell lines) -Proteomics metabolics ( binding assays to identify target interactions |
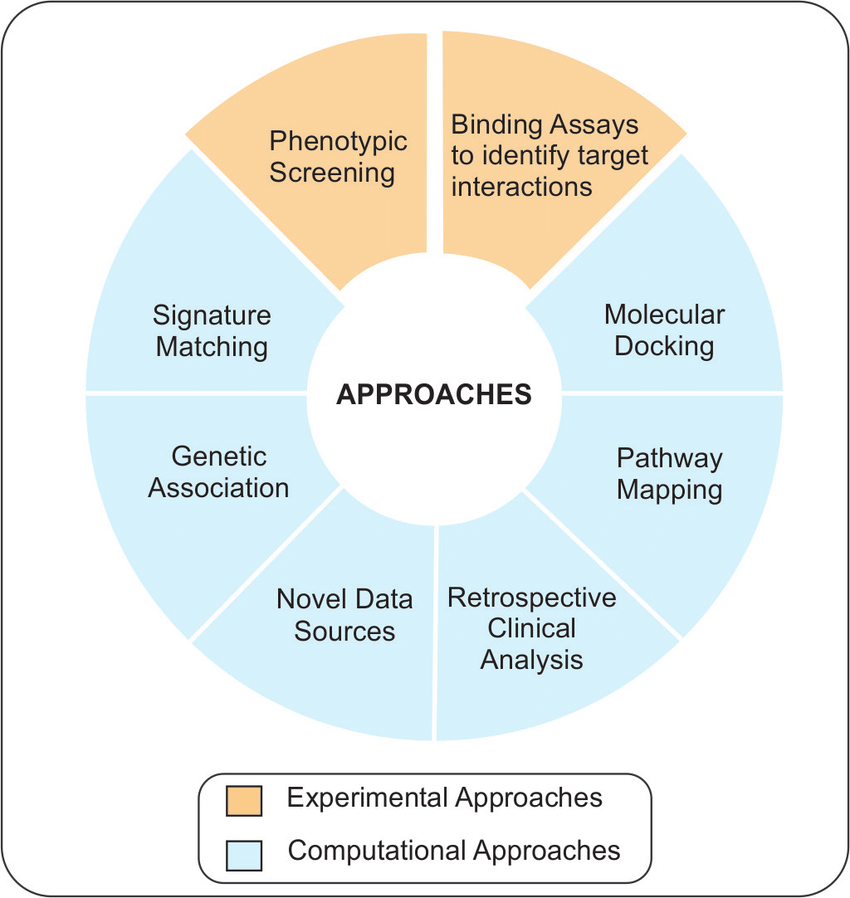
Figure 2: Approaches to drug repurposing
Challenges of drug repurposing
- Technical challenges: Pharmaceutical organizations may face several technical challenges to drug repurposing as mentioned below;
- Data volume and hygiene: access to great volumes of data (patent data, pharmacological data, compound libraries and published scientific literature as required for drug repurposing. Such huge data suffer from poor data hygiene, the probability of errors and duplications. Moreover, the datasets might be restricted due to privacy or for security matters.
- Data heterogeneity: different types of data originating from multiple sources and/or stored in multiple formats. This poses significant challenges for analysis, management, and integration.
- Lack of technical experience: a cross-domain expertise that follows science and technology is required to create a knowledge graph to organize complex biological data into a network.
- Efficacy and safety: a repositioned drug may show lower effectiveness for a new indication than it was for the original licensed indication. Also, candidates for drug repurposing may not be as beneficial when they are newly employed in combination therapeutic indications than in their former single therapy.
- Regulatory challenges: Regulatory considerations are critical attributes for the development of repurposed drug approaches.
- Organisational barriers in industry and intellectual property rights: repurposing in the pharmaceutical industry can be encountered with one or two organizational hurdles, especially if the repurposed pharmacological indication is not within the core disease area of the organization or the compound is no longer under development (no ‘live’ project within the R&D section) to provide purposeful support for the new indication. Moreover, the achievement of new licensing agreements for a repositioned drug can be a complicated and time-consuming process. If a pharmaceutical organization owns a drug whose patent is near-expiry, they might likely try to extend the patent protection if a new therapeutic indication is discovered.
Strategies of drug repurposing
Identifying novel biological indications of some FDA approved drugs has been a practicable strategy to speed up the drug discovery process.
- On-target drug repurposing: where the known pharmacological mechanism of a certain drug molecule is applied to a new therapeutic indication. To explain; the biological target of that drug compound is same, but the disease is different. An example for that is minoxidil which is primarily an antihypertensive vasodilator, so is newly used to treat male baldness.
- Off-target drug repurposing: where the pharmacological mechanism is unknown, and so drug candidates act on new targets, outside of their primary scope, for a new therapeutic indication. The target and the indications are marked as a novel approach. An example of that is aspirin, which is originally used in treating different pain and inflammatory disorders. Aspirin also suppresses blood coagulation and is therefore used in the treatment of heart attacks. In addition, aspirin has been recently used in the treatment of prostate cancer. Figure 3 briefly explains both strategies.
Some success stories for repurposed drugs:
A. Drug repurposing in oncology
Clinically efficacious non-cancer drugs that are known to target a homologous biological mechanism or mutation can be effectively repositioned to treat various forms of cancer.
| Drug, pharmacological category | Approved indication | New indication | Successful repositioning from FDA FDA-approved drug |
| Aspirin, NSAID | Pain & inflammation | Prostate cancer | Successful repositioning of FDA FDA-approved drug |
| Celecoxib, NSAID | inflammation | Breast & colon cancer | Successful repositioning of an FDA-approved drug |
| Cimetidine, h2receptor antagonist | Gastric ulcer | Breast, lung, prostate cancer | Successful repositioning of an FDA-approved drug |
| Daunorubicin antibiotic | Breast cancer | Successful repositioning of an FDA-approved drug | |
| Digoxin, cardiotonic | Heart failure | Prostate cancer | Successful repositioning of an FDA-approved drug |
| disulfiram | Chronic alcoholism | cancer | Successful repositioning of an FDA-approved drug |
| Itraconazole, antifungal | Fungal infections | cancer | Successful repositioning of an FDA-approved drug |
| mebendazole | antiparasitic | Brain cancer | Successful repositioning of an FDA-approved drug |
| metformin | diabetes | Successful repositioning of FDA FDA-approved drug | Successful repositioning of an FDA-approved drug |
| orlistat | obesity | cancer | Successful repositioning of an FDA-approved drug |
| Ribavirin, antiviral | hepatitis | Leukaemia, lymphoma | Successful repositioning of FDA-approved drug |
| Statins, HMG-CoA reductase inhibitors | Anti-hypercholesterolaemia | cancers | Successful repositioning from FDA-approved drug |
Table 1: Some examples of promising re-positioning drugs in oncology
B- Drug repurposing during a public health crisis
During the COVID-19 pandemic, various drugs were candidates for drug repurposing under emergency authorization. Remdesivir, being an antiviral, and dexamethasone, as a widely used steroid, were the top candidates. The following table illustrates more candidates for drug repositioning
| Drug, pharmacological category | Approved indication | New indication | Status of development |
| Azithromycin, antibacterial | Upper respiratory Bacterial infections | COVID-19 | Successful repositioning of an FDA-approved drug |
| Chloroquine, antimalarial | malaria | COVID-19 | Ivermectin, an anthelmintic |
| Colchicine, an anti-inflammatory | Gout | COVID-19 | Successful repositioning of FDA-approved drug |
| Favipiravir, antiviral | influenza | COVID-19 | Successful repositioning of an FDA-approved drug |
| Hydroxychloroquine, antimalaria | Malaria, Rheumatoid arthritis | COVID-19 | Successful repositioning of an FDA-approved drug |
| Ivermectin, anthelmintic | Scabies, helminthiasis | COVID-19 | Successful repositioning of an FDA-approved drug |
| Remdesivir, antiviral | Influenza, Ebola | COVID-19 | Successful repositioning from investigational new drug |
| Tocilizumab, IL-6 inhibitor | Rheumatoid arthritis | COVID-19 | Successful repositioning from an FDA-approved drug |
Table 1: Some examples of promising re-positioning drugs in oncology
C- Drug repurposing for rare diseases
Collaborative approaches strengthen scientific and technical expertise to initiate development, acknowledged examples of repurposing for rare diseases. The best-known example is the identification of four new candidates for drug reprofiling to treat chronic pancreatitis.
Conclusion
Targeting the development of good therapeutic options for various diseases is challenging. Recently, drug repurposing has brought a new approach towards the development of new therapeutic indications based on existing and/or approved medicines. Repurposing a drug is one concept towards the detailed investigation of the likely therapeutic potential of different molecules. Higher success rates of repositioned drugs are attained through thorough knowledge, along with integrated approaches between computational and experimental methods.
References
- Drug repurposing: an effective tool in modern drug discovery, feb 2023
- Drug discovery today, 2014
- Drug repurposing: an emerging approach in drug discovery, 2020
- Drug repurposing: approaches and methods
- Repurposing drugs in oncology, 2015
- Drug repositioning: identifying and developing new uses for existing drugs, 2005
- Drug repurposing: progress, challenges and recommendations, 2019
Dive Deeper into Preformulation Studies
Understanding the solubility and stability of a drug is crucial in the early stages of pharmaceutical development. To explore these aspects in detail, read our comprehensive guide:
👉 Preformulation Study Part 2: Solubility, Stability & Key Parameters in Drug Development