Emulsion stability is a critical aspect in pharmaceutical, cosmetic, and food formulations, ensuring the uniformity and effectiveness of products.
Scientifically, we will talk about emulsion instability problems and study the physical issues and root causes of each problem.
Emulsion definition
Emulsion is a word coming from emulsifier which can help two phases to be like a homogenous phase by decreasing the surface tension between the two phases for dedicated features needed for medical or cosmetic use. Emulsion stability is the key indicator for good emulsion formulation.
An emulsion is a mixture of two immiscible liquids, stabilized by an emulsifier that reduces surface tension and ensures a uniform blend.
Continuous Phase vs. Dispersed Phase
Emulsions consist of two primary phases:
| Continuous phase | Dispersed phase |
|---|---|
| -This is the phase in which the other phase is dispersed. -It forms the bulk of the surrounding medium of the emulsion. For example: – In an oil-in-water (O/W) emulsion, water is the continuous phase. – In a water-in-oil (W/O) emulsion, oil is the continuous phase. | -This is the internal phase, consisting of tiny droplets of a liquid distributed within the continuous phase. -It influences the functional properties of the emulsion, such as drug release in pharmaceuticals or moisturizing effects in cosmetics. For example: – In an oil-in-water (O/W) emulsion, oil is the dispersed phase. – In a water-in-oil (W/O) emulsion, water is the dispersed phase. |
Emulsifiers definition
An emulsifier is a substance that helps mix two immiscible liquids, such as oil and water, by reducing surface tension and stabilizing the emulsion. Emulsifiers are essential in pharmaceuticals, cosmetics, and food industries, ensuring uniform texture, stability, and enhanced product performance.
Types of Emulsifiers
Emulsifiers can be classified based on their chemical nature and function:
Types according to charge
| Ionic Emulsifiers | Non-ionic Emulsifiers | Zwitterionic Emulsifiers |
|---|---|---|
| -Charged emulsifiers. -Either positive (Cationic) or negative (Anionic). These emulsifiers stabilize the emulsion by creating electrostatic repulsion between droplets of the dispersed phase. | -Non-charged emulsifiers. -These emulsifiers stabilize emulsions by forming steric barriers around the droplets, preventing coalescence. | -Carrying both positive and negative charges on the same molecule. -Electrically neutral. -They adapt their charge based on the pH of the surrounding medium. |
| Examples: – Anionic emulsifiers: Sodium stearate and Magnesium stearate. – Cationic emulsifiers: Quaternary ammonium compounds (e.g., cetyltrimethylammonium bromide). | Examples: Polysorbates (e.g., Tween 80) and sorbitan esters (e.g., Span 60). | Examples: Lecithin and phospholipids. |
Types according to nature
| Natural Emulsifiers | Semi-Synthetic Emulsifiers | Synthetic Emulsifiers |
|---|---|---|
| -Derived from natural sources such as animals, plants, or microorganisms. -They are biodegradable, biocompatible, non-toxic, and safe for use in pharmaceutical, food, and cosmetic products. | -Chemically modified derivatives of natural substances. -Designed to enhance their emulsifying properties or improve stability. | -Chemically synthesized. -Provide high efficiency and emulsion stability. -Highly versatile and can be tailored for specific formulations. |
| Examples: Lecithin, Acacia Gum, and Pectin. | Examples: Hydroxypropyl methylcellulose (HPMC), Carboxymethyl cellulose (CMC), and Modified starches. | Examples: Polysorbates (e.g., Tween 20, Tween 80), Sorbitan Esters (e.g., Span 20, Span 80), and Sodium Lauryl Sulfate (SLS). |
In this article, we talk about physical mechanisms for emulsion breakdown, which would happen at the emulsion formula
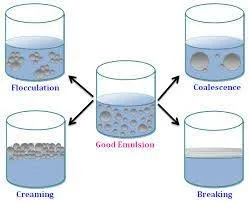
Mechanism of emulsion breakdown
Mechanism of emulsion breakdown:
physical
- Creaming and sedimentation:
- Coalescence and Flocculation
- Ostwald reponing
- Phase inversion
- breaking (Cracking)
Chemical
Creaming and Sedimentation:
- Affect emulsion stability.
- Creaming is upward creaming that happens mainly in O/W emulsions due to density difference and gravitational force, which is oil phase density being lower than water phase density, but there is no change in droplet size and could be reversible, for example (Milk).
- Unlike, sedimentation which is down-ward creaming that happened mainly in W/O emulsions Due to density difference and gravitational force, which is oil phase density being higher than water phase density.
How can it be prevented?
Prevention action for this problem, which may affect emulsion stability, we could target decreasing the droplet size and also, decreasing the difference between the two phases’ density, and increasing the continuous phase in each emulsion according to the type of emulsion.
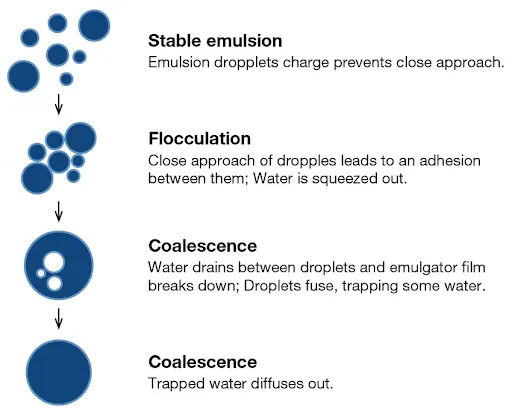
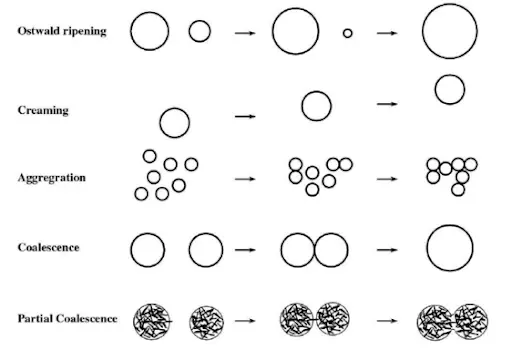
Flocculation (Aggregation) and Coalescence
- Affect emulsion stability.
- Flocculation is reversible and could happen by aggregation of two or more droplets without changing their individual density, but the root cause here is the attractive forces which is higher than repulsive forces, the balance between these forces changing due to PH changing and also, ionic strength of aqueous medium.
- Also, increasing the emulsifier concentration or adding a high HLB (Hydrophilic Lipophilic Balance) emulsifier.
- Flocculation is the first step for coalescence, which is irreversible (Breaking the interfacial film surrounding the water globule in the case of W/O emulsion).
- Extensive coalescence can lead to the formation of separate layers on top of the product, which is known as oiling off.
- This may happen due to many factors:
1- Incompatibility of adding substances with the emulsifying agent.
2- Temperature change.
3- Addition of an excessive amount of dispersed phase.
the major factor that can prevent coalescence is the mechanical strength of the interfacial film.
How can it be prevented?
- Using Proper Emulsifiers.
- Particle size control.
- Electrostatic Stabilization.
- Steric Stabilization.
- Viscosity Control.
- pH and Ionic Strength Optimization.
- Temperature control.
- Avoiding Mechanical Stress
Ostwald ripening
Ostwald ripening is the growth of one emulsion droplet of the dispersed phase at the expense of a smaller one as a result of the difference in the chemical potential of the material within the droplets.
How can it be prevented?
- Use of Stabilizers and Emulsifiers.
- Reduce Droplet Size.
- Increase the Viscosity of the Continuous Phase.
- Optimize the Oil-to-Water Ratio.
- Control Temperature.
- Addition of Anti-Ripening Agents.
- Use of Coalescence Inhibitors.
- Addition of Higher Concentration of Dispersed Phase.
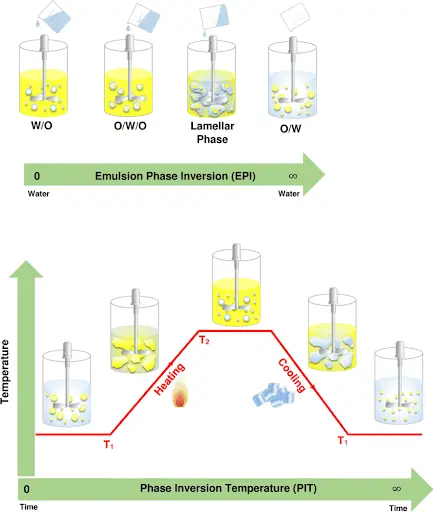
Phase inversion
Phase inversion is the switching of an emulsion system to another emulsion system (O/W to W/O Or vice versa) which is happening due to many factors :
1- Wrong emulsifying agents.
2- Addition of electrolytes.
3- Temperature changes.
4- Changing phase volume ratio.
Breaking (Cracking)
Breaking is the separation of an emulsion into two phases: the continuous phase and the dispersed phase, which is irreversible due to:
1- Addition of an emulsifying agent of opposite type.
2- Decomposition or precipitation of the emulsifying agent.
3- Increasing temperature.
4- Freezing.
5- Addition of common solvent.
How can it be prevented?
- Absence of incompatibility between the emulsifier and other ingredients.
- Avoid an excessive amount of dispersed particles.
- Avoid disturbance at temperatures by increasing or freezing.
- Avoid microorganism growth.
General notes affecting emulsion stability
- Keeping the concentration of the dispersed phase between 30-60 % to prevent phase inversion.
- Light and rancidity influence the color and odor of oils and may destroy their vitamin activity